
Blog
7 A Level Chemistry Exam Mistakes You’re Probably Making (And How to Fix Them)

You’ve put in the hours. You’ve made flashcards, watched videos, and filled notebooks with equations. So why aren’t your grades reflecting all that hard work?
Here’s the thing , A Level Chemistry isn’t just about knowing the content. It’s about knowing how to communicate that knowledge in the exact way examiners expect. And that’s where so many students get caught out.
I see it all the time working as an A Level Chemistry tutor. Bright, hardworking students losing marks not because they don’t understand the chemistry, but because of small, fixable mistakes that add up fast.
The good news? Once you know what these mistakes are, you can start avoiding them immediately. Let’s walk through the seven most common exam errors I see , and more importantly, how to fix them. 🌟
1. Misreading the Question (Those Sneaky Command Words)
This one costs students so many marks, and it’s heartbreaking because they often know the answer , they just don’t give the right type of answer.
Here’s the problem: “Describe” and “Explain” are not the same thing. Neither are “State,” “Suggest,” “Compare,” or “Justify.” Each command word tells you exactly what the examiner wants, and if you ignore it, you’re essentially answering a different question.
Describe = Say what happens (the “what”)
Explain = Say what happens AND why it happens (the “what” + “because”)
For example, if a question asks you to describe the trend in atomic radius across Period 3, you just need to say it decreases. But if it asks you to explain, you need to add that nuclear charge increases while shielding stays roughly the same, so electrons are pulled in more strongly.
The fix: Before you write anything, underline the command word and ask yourself: “What type of answer is this asking for?” Take five seconds to do this on every single question. It sounds simple, but this habit alone can rescue your grade.

2. Forgetting State Symbols in Equations
This might seem like a tiny detail, but state symbols are often worth a mark , and examiners are specifically looking for them.
When a question asks you to write an equation, check whether it says “include state symbols.” If it does and you leave them out, that’s a mark gone. Even if your equation is perfectly balanced.
The four state symbols you need:
- (s) = solid
- (l) = liquid
- (g) = gas
- (aq) = aqueous (dissolved in water)
The fix: Make it a non-negotiable habit. Every time you write an equation in practice, include state symbols automatically. That way, it becomes second nature by exam day. Your future self will thank you! 🙌
3. Unit Conversion Errors (The Ideal Gas Law’s Favourite Victim)
If there’s one area where I see students lose marks consistently, it’s unit conversions , especially in calculations involving the Ideal Gas equation (pV = nRT) and thermodynamics.
The Ideal Gas constant R = 8.314 J K⁻¹ mol⁻¹ requires:
- Pressure in Pascals (Pa), not kPa
- Volume in m³, not cm³ or dm³
- Temperature in Kelvin, not °C
Getting even one of these wrong throws off your entire answer. And because examiners can only award method marks if your working is clear, a conversion error right at the start can cost you nearly all the marks on a question.
The fix: Write out your unit conversions as a separate, visible step before plugging numbers into the equation. For example:
- 25°C → 298 K ✓
- 500 cm³ → 0.0005 m³ ✓
- 101 kPa → 101,000 Pa ✓
This takes seconds and makes your working crystal clear. If you’d like more help with this, I’ve written a whole post on how to tackle calculations without panic that you might find useful.
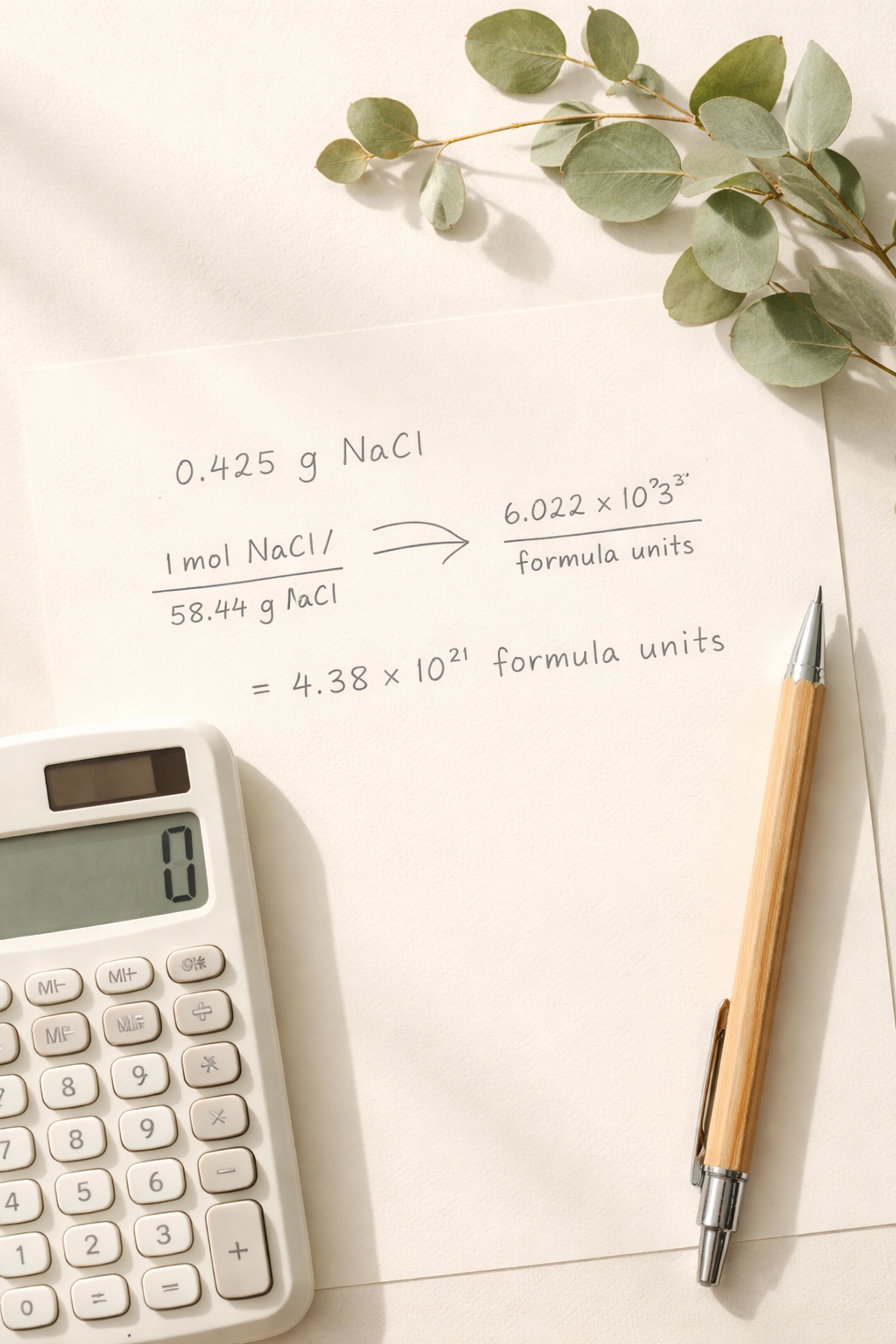
4. Not Showing Enough Working Out
Speaking of working out , you need to show it. All of it.
I know it feels faster to do steps in your head, especially when you’re confident. But here’s what happens: you make one small slip, get the wrong final answer, and the examiner can’t see where you went wrong. Result? Zero marks instead of the three or four you could have salvaged.
Mark schemes are designed to award method marks for correct steps, even if your final answer is wrong. But they can only do this if your working is visible and logical.
The fix: Treat every calculation like you’re explaining it to someone else. Write out:
- The formula you’re using
- The values you’re substituting (with units)
- Each step of the calculation
- Your final answer with the correct units and appropriate significant figures
Yes, it takes a bit longer. But it’s worth it : both for picking up method marks and for catching your own errors before you move on.
5. Confusing Intermolecular Forces with Covalent Bonds
This one trips up even strong students, and it’s a classic examiner trap.
When a question asks why water has a higher boiling point than methane, they want you to talk about intermolecular forces : the forces between molecules. They do NOT want you to talk about covalent bonds, which are the forces within molecules.
Here’s the key distinction:
- Covalent bonds hold atoms together inside a molecule (strong, don’t break during boiling)
- Intermolecular forces hold molecules together (weaker, these ARE what break during boiling)
When you boil water, you’re overcoming hydrogen bonds between H₂O molecules, not breaking O-H covalent bonds.
The fix: Whenever a question mentions melting points, boiling points, or physical properties, your brain should immediately flag: “This is about intermolecular forces.” Get into the habit of using precise terminology : hydrogen bonding, permanent dipole-dipole, and London dispersion forces : depending on the molecules involved.

6. Missing the “Per Mole” in Enthalpy Changes
Enthalpy questions are generous with marks if you get them right : and brutal if you make this common mistake.
When you calculate or define an enthalpy change, it must be expressed per mole of a specific substance. Standard enthalpy of combustion? That’s the enthalpy change when one mole of a substance burns completely. Standard enthalpy of formation? The enthalpy change when one mole of a compound forms from its elements.
If your equation shows 2 moles being burned and you don’t adjust your final answer, you’ve just halved (or doubled) your marks away.
The fix: Always check: “Is my answer per mole?” If your equation involves 2 moles of the substance, divide your calculated enthalpy by 2. And when writing definitions, always include “per mole” : examiners look for this phrase specifically. ❤️
7. Poor Time Management on Multi-Part Questions
Finally, let’s talk about time : because running out of it is one of the most stressful things that can happen in an exam.
Multi-part questions (the ones worth 10+ marks) are particularly dangerous. Students often spend too long perfecting parts (a) and (b), then rush through (c) and (d) or leave them blank entirely. But here’s the thing: part (d) is often worth the most marks.
The fix: Use the mark allocation as your guide. If a question is worth 3 marks, spend roughly 3-4 minutes on it. If it’s worth 6 marks, you can afford more time : but not 15 minutes.
During practice, time yourself religiously. Get comfortable with the feeling of moving on before you’re 100% happy with an answer. A good answer on every question beats a perfect answer on half of them.
Ready to Close the Gap Between Effort and Results?
These mistakes are fixable. Every single one of them. And once you start catching them in practice, you’ll be amazed at how quickly your marks improve.
If you’re studying GCSE Chemistry or A Level and want someone in your corner to help you build these habits, I’d love to help. At Chemistry with Chloe, I offer 1-1 and small group tuition tailored to your exam board and your goals. Whether you’re based in the UK, Dubai, or anywhere else in the world, we can work together online at times that suit you.
I also support students with medical school applications, including UCAT preparation and personal statement guidance : because I know how important that Chemistry grade is for your future. You can read more about why Chemistry excellence matters for aspiring doctors right here.
Ready to work smarter, not just harder? Get in touch ( I’d love to hear from you.) 🌟
Oxford-Educated Chemistry Specialist
With over 20 years of teaching experience at some of the UK’s top independent schools, I help ambitious students bridge the gap between hard work and top-tier results. I specialise in GCSE, A Level, and IB Chemistry tuition for students targeting Grade 9s and A*s. Based in the UK but working globally, I provide 1-1 online support for families in South and West London, Dubai, and Hong Kong, ensuring students are perfectly prepared for competitive medical applications and Oxbridge entries.
I’ve helped students achieve top grades from schools such as Alleyn’s, Dulwich College, Tonbridge, Sevenoaks, Brighton College, Wycombe Abbey, Caterham, St Paul’s, Dubai College, Dubai British School and Harrow International School Hong Kong.
Contact me archardchloe@gmail.com to discuss how I can help your child excel in Chemistry.
