
Edexcel A Level Chemistry Core Practicals 1-16
Core Practical 1: Measuring Molar Volume (Catching Bubbles like a Pro)
Welcome to your first major hurdle in the world of Edexcel A Level Chemistry practicals! If you’ve spent any time in a lab recently, you know that things rarely go as smoothly as the textbook says they will. Chemicals spill, stoppers pop off, and gas bubbles have a frustrating habit of disappearing into thin air.
Core Practical 1 is all about Molar Volume. Specifically, we are trying to find out exactly how much space one mole of a gas occupies. According to the theory (shoutout to Avogadro), one mole of any gas at Room Temperature and Pressure (RTP) should take up about 24 dm³ (or 24,000 cm³).
But does it really? That’s what we’re here to find out. Grab your goggles, and let’s dive into the bubbly world of Edexcel Core Practical 1.
The Big Idea: What are we actually doing?
In this experiment, we react a known mass of Sodium Carbonate (Na2CO3) with an excess of Hydrochloric Acid (HCl).
The reaction looks like this:
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)
Our mission is simple:
- Weigh the carbonate.
- React it with the acid.
- Catch all the carbon dioxide gas produced.
- Use the volume of that gas to calculate the molar volume.
If we know how many moles of Na2CO3 we started with, we know how many moles of CO2 should be produced (it’s a 1:1 ratio). By measuring the actual volume of that CO2, we can work out the volume per mole.
The Kit: Choosing Your Weapon
There are two main ways to catch these bubbles. Your teacher might let you choose, or you might be tested on the pros and cons of both in your Paper 3 exam.
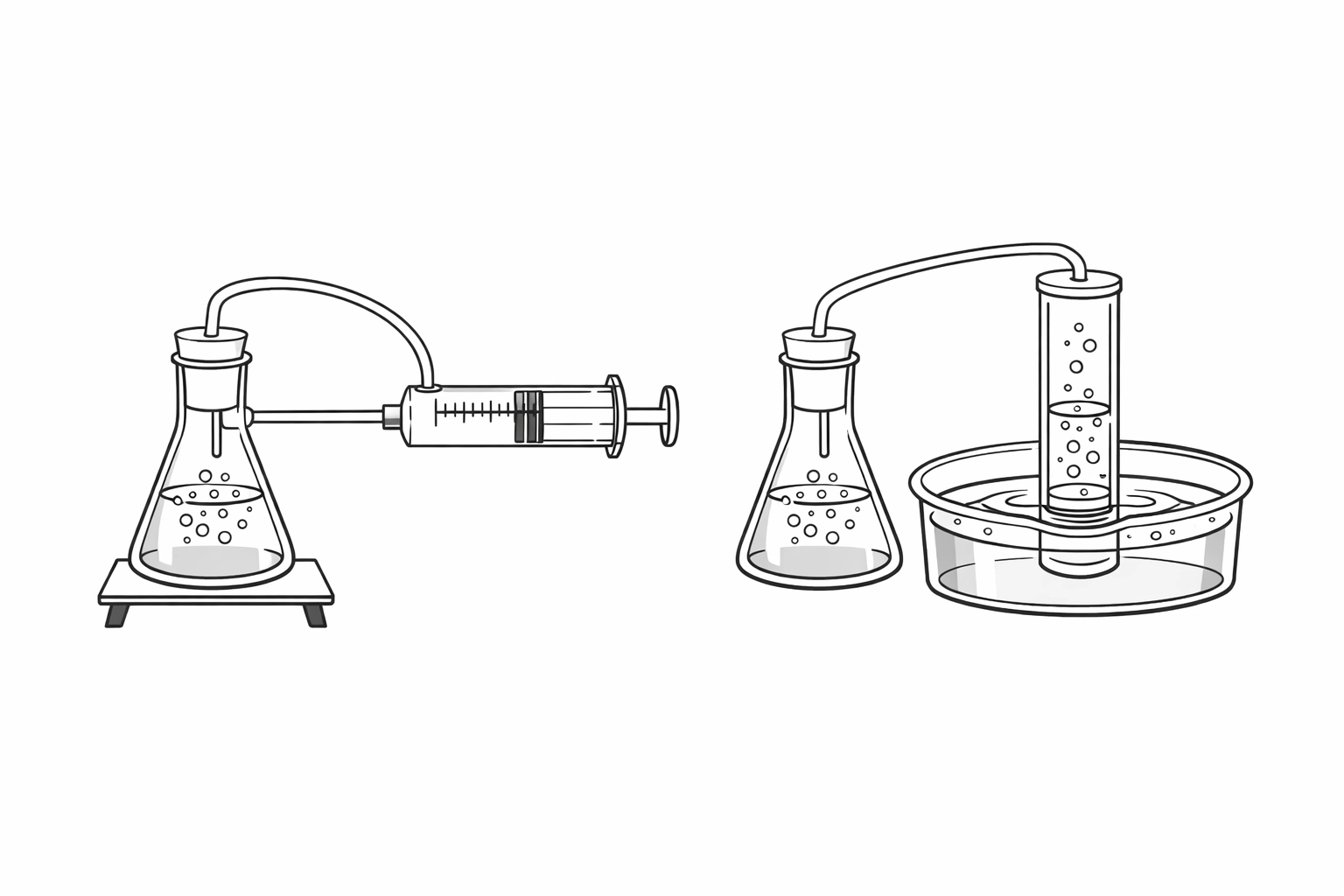
Option A: The Gas Syringe
This is the “cleanest” method. You connect a sealed conical flask to a gas syringe via a delivery tube. As the gas is produced, it pushes the plunger out.
- Pros: Very accurate; no risk of the gas dissolving in water.
- Cons: Syringes can be “sticky” if they aren’t clean, leading to pressure build-up and leaks.
Option B: Displacement of Water
This is the classic “bubbles in a tube” setup. You run a delivery tube from your flask into a trough of water and up into an inverted measuring cylinder (or a burette) filled with water.
- Pros: You get to play with water; very visual.
- Cons: CO2 is slightly soluble in water, so you might “lose” some gas before you can measure it.
The Method: Step-by-Step
To get top marks, you need to be precise. Here is the “Golden Path” for this practical:
- The Weighing Trick: Use the weighing by difference method. Weigh a sample bottle containing your Na2CO3. Tip the powder into your conical flask. Re-weigh the empty bottle. Subtract the two to find the exact mass of carbonate used. This is much more accurate than just zeroing the balance.
- The Acid Prep: Measure out an excess of HCl (usually about 30 cm³ of 1.0 mol dm⁻³ acid). “Excess” is a keyword here: it ensures all the carbonate reacts.
- The Great Gas Escape: This is the tricky part. You need to add the acid to the flask and replace the bung instantly. Any delay means CO2 escapes into the room instead of into your measuring device.
- Pro Tip: Some setups use a small sample tube of acid inside the flask that you tip over once the bung is sealed. This is the “Boss Level” way to prevent gas loss.
- Wait for the Fizz: Wait until the effervescence (bubbling) stops. Give the flask a gentle swirl to make sure every last bit of carbonate has met its acidic doom.
- Record: Note the final volume of gas.
The Math Part (Don’t Panic)
Once you have your data (Mass of Na2CO3 and Volume of CO2), it’s time for the calculations.
Step 1: Find the moles of Na2CO3
n = mass / molar mass (Mr)
(The Mr of Na2CO3 is 106.0)
Step 2: Find the moles of CO2
Because the ratio is 1:1, the moles of CO2 is the same as the moles of Na2CO3.
Step 3: Calculate Molar Volume
Molar Volume = Volume of gas (measured) / Moles of gas (calculated)
Make sure your units match! If your volume is in cm³, your answer will be in cm³ mol⁻¹. To get it into the standard dm³ mol⁻¹, divide by 1000.
The “Boss Level” Application: Finding the Unknown Metal
Examiners love to twist this practical. Instead of Na2CO3, they might give you an “unknown metal carbonate” with the formula MCO3.
They will give you the mass and the volume of gas produced. Your job is to:
- Calculate the moles of gas (using the molar volume of 24,000 cm³ mol⁻¹).
- Know that Moles of Carbonate = Moles of Gas.
- Calculate the Mr of the carbonate: Mr = mass / moles.
- Subtract the Ar of Carbon and Oxygen (12 + 16 × 3 = 60).
- The remaining number is the Ar of your metal M. Look at your Periodic Table: who is it? (If it’s around 40, it’s Calcium!).
Why Is My Data Bad? (Common Errors)
If your calculated molar volume isn’t exactly 24 dm³, don’t worry! Here are the reasons why, which you’ll likely need to write about in your evaluation:
- Gas Leakage: The most common error. If you aren’t fast with the bung, the volume will be too low.
- CO2 Solubility: As mentioned, CO2 dissolves in water. Using a gas syringe or saturating the water with CO2 beforehand helps fix this.
- Temperature & Pressure: Molar volume is highly sensitive to the room’s conditions. If the lab is freezing or you’re on top of a mountain, the results will shift.
- Incomplete Reaction: If your acid wasn’t strong enough or you didn’t swirl the flask, some carbonate might stay unreacted at the bottom.
Safety First
A-Level Chemistry isn’t just about blowing things up; it’s about not hurting yourself while you do it.
- HCl is an Irritant: At 1.0 mol dm⁻³, it’s not super dangerous, but it will sting if it gets in a cut or your eyes. Wear goggles.
- Glassware: Gas syringes are expensive and fragile. Don’t let them roll off the bench!
- Pressure: Never block the end of the delivery tube or the syringe. You’re building up gas pressure; it needs somewhere to go, or something will pop.
Summary Checklist for Revision
Before you head into your exam, make sure you can:
- [ ] Draw the apparatus for both gas collection methods.
- [ ] Explain why “weighing by difference” is better than just taring the balance.
- [ ] Write the balanced equation for the reaction.
- [ ] Convert cm³ to dm³ without breaking a sweat.
- [ ] Identify two ways to reduce the percentage uncertainty in the volume measurement (e.g., using a larger mass of carbonate to produce a larger volume of gas).
Core Practical 1 is the foundation for all the gas calculations you’ll do in Year 12 and 13. Master the bubbles now, and the rest of physical chemistry will feel like a breeze!
What’s next? If you’ve nailed Molar Volume, you might want to check out our guide on Edexcel A Level Chemistry Topic 2: Bonding and Structure or move on to the next practical: Titrations!
Happy experimenting! 🧪✨

