
Edexcel A Level Chemistry Topic 10: Equilibrium I

Equilibrium I: The Great Balancing Act (Mastering Kc & Le Chatelier).
When you first start Chemistry, you’re often taught that reactions go from A to B. You mix two things together, they fizz, and you get a product. But as you move into Edexcel A Level Chemistry, you start to realise that the chemical world isn’t always a one-way street. Many reactions are actually a two-way conversation.
Welcome to the world of equilibrium. In this guide, we’re breaking down Topic 10: Equilibrium I. We’ll look at why some reactions “stuck” in the middle, how we can bully them into making more product, and how to write the mathematical expressions that define these balances.
What is Dynamic Equilibrium?
Imagine a busy shop. People are walking in through the front door, and people are walking out through the back door. If people are entering at exactly the same rate they are leaving, the total number of people inside the shop stays the same. To an observer outside, it looks like nothing is changing, but inside, there is constant movement.
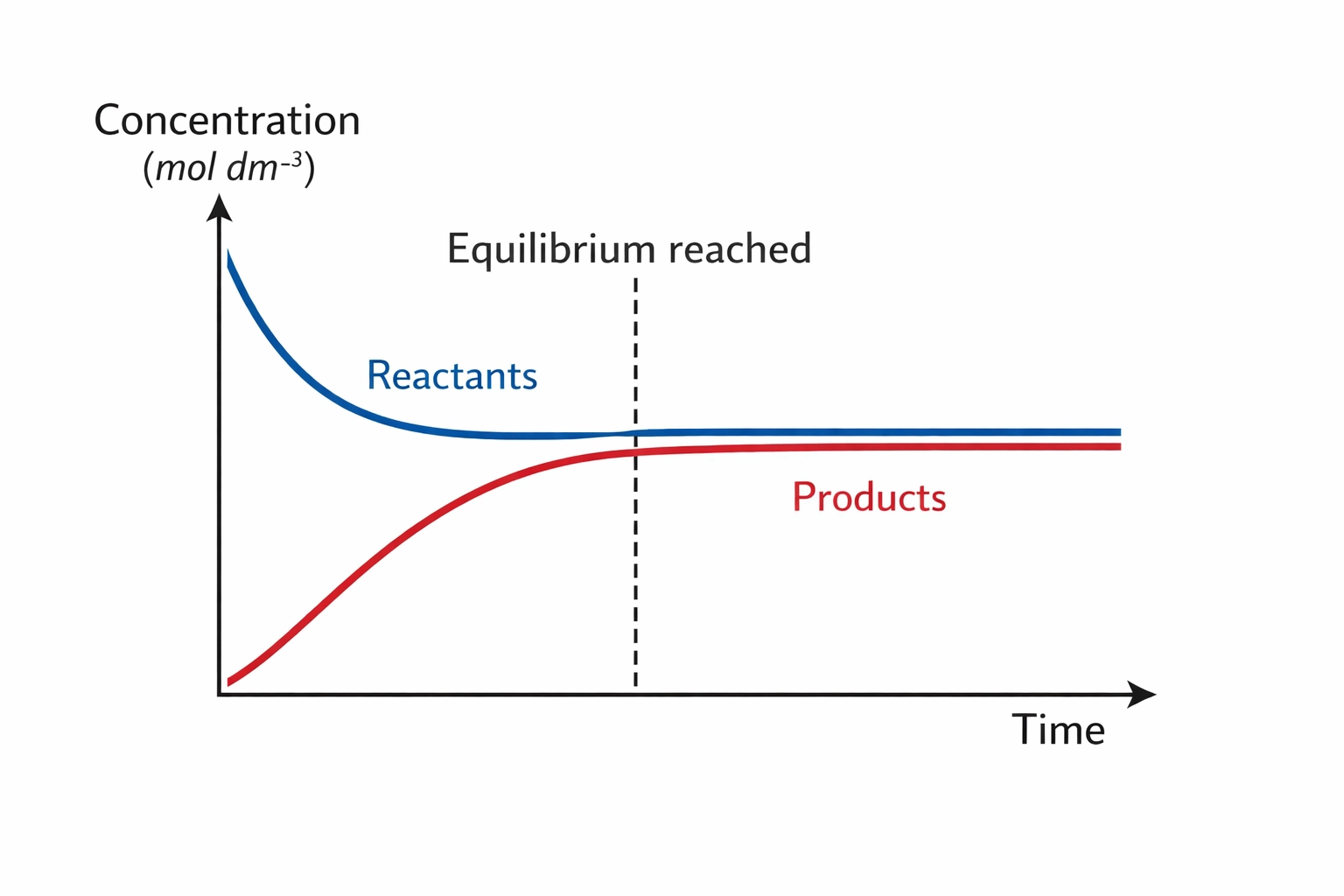
This is exactly what dynamic equilibrium is. In a reversible reaction (represented by the double arrow ⇌), the forward and backward reactions are happening at the same time.
For a system to be in a state of dynamic equilibrium, it must meet two strict criteria that examiners love to ask about:
- The rate of the forward reaction is equal to the rate of the backward reaction.
- The concentrations of reactants and products remain constant.
It is vital to remember that “constant” does not mean “equal.” You might have 90% product and 10% reactant, or vice versa. As long as those percentages aren’t changing anymore, you’re at equilibrium. Also, this can only happen in a closed system, meaning nothing can escape or enter (like gases floating away into the lab).
Le Chatelier’s Principle: The Reaction’s Rebellion
Once a reaction has reached its happy place (equilibrium), it really doesn’t like being disturbed. Henri Louis Le Chatelier came up with a principle to describe this “grumpiness.”
Le Chatelier’s Principle states: If a system at equilibrium is disturbed by a change in conditions, the system will shift its position to counteract that change.
Essentially, the reaction tries to undo whatever you just did. Let’s look at the three main ways we can disturb a system.
1. Changing Concentration
If you add more reactant to a system, the equilibrium thinks, “Ugh, too much reactant. I need to get rid of it.” It shifts to the right (the product side) to use up that extra reactant.
If you remove a product as it’s formed, the equilibrium thinks, “Wait, where did my product go? I need to make more!” and it shifts to the right again. This is a classic trick used in industry to keep reactions moving forward.
2. Changing Pressure
This only affects reactions involving gases. When you increase the pressure, the system tries to lower it. How? By shifting to the side with fewer moles of gas.
Take the production of ammonia:
N2(g) + 3H2(g) ⇌ 2NH3(g)
On the left, we have 4 moles of gas (1 + 3). On the right, we have 2 moles. If we turn up the pressure, the equilibrium shifts to the right because 2 moles of gas exert less pressure than 4 moles.
3. Changing Temperature
This is the one that trips students up the most. To predict the shift, you must know if the forward reaction is exothermic (gives out heat, negative enthalpy) or endothermic (takes in heat, positive enthalpy).
- Increase Temperature: The system wants to cool down. It shifts in the endothermic direction to “absorb” the extra heat.
- Decrease Temperature: The system wants to warm up. It shifts in the exothermic direction to release heat.
Pro Tip: Temperature is the only factor that actually changes the numerical value of the equilibrium constant (Kc). Changes in concentration or pressure might shift the position, but Kc stays the same.
The Industrial Balancing Act: Yield vs. Rate
In a perfect world, industrial chemists would get a 100% yield of product instantly. In the real world, we have to deal with a massive compromise between yield and rate.
Let’s look at a generic exothermic reaction where we want to make product.
- To get a high yield, Le Chatelier says we should use a low temperature (because the reaction is exothermic).
- However, at a low temperature, the particles have less kinetic energy, meaning fewer successful collisions. The rate of reaction would be painfully slow. It might take years to get that high yield!
So, chemists choose a compromise temperature. It’s high enough to get the product made quickly, but low enough that we still get a decent amount of it.
The same goes for pressure. High pressure usually increases both yield and rate for many gaseous reactions, but building pipes and tanks that don’t explode under high pressure is incredibly expensive and dangerous. Therefore, a compromise pressure is used to balance safety, cost, and output.
Deducing the Equilibrium Constant (Kc)
While Le Chatelier gives us a qualitative “vibe” of where the reaction is going, Kc gives us the hard numbers. Kc is the ratio of product concentrations to reactant concentrations at equilibrium.
For a general reaction:
aA + bB ⇌ cC + dD
The expression is:
Kc = [C]c [D]d / [A]a [B]b
(Remember: Square brackets [ ] always mean “concentration in mol dm-3”.)
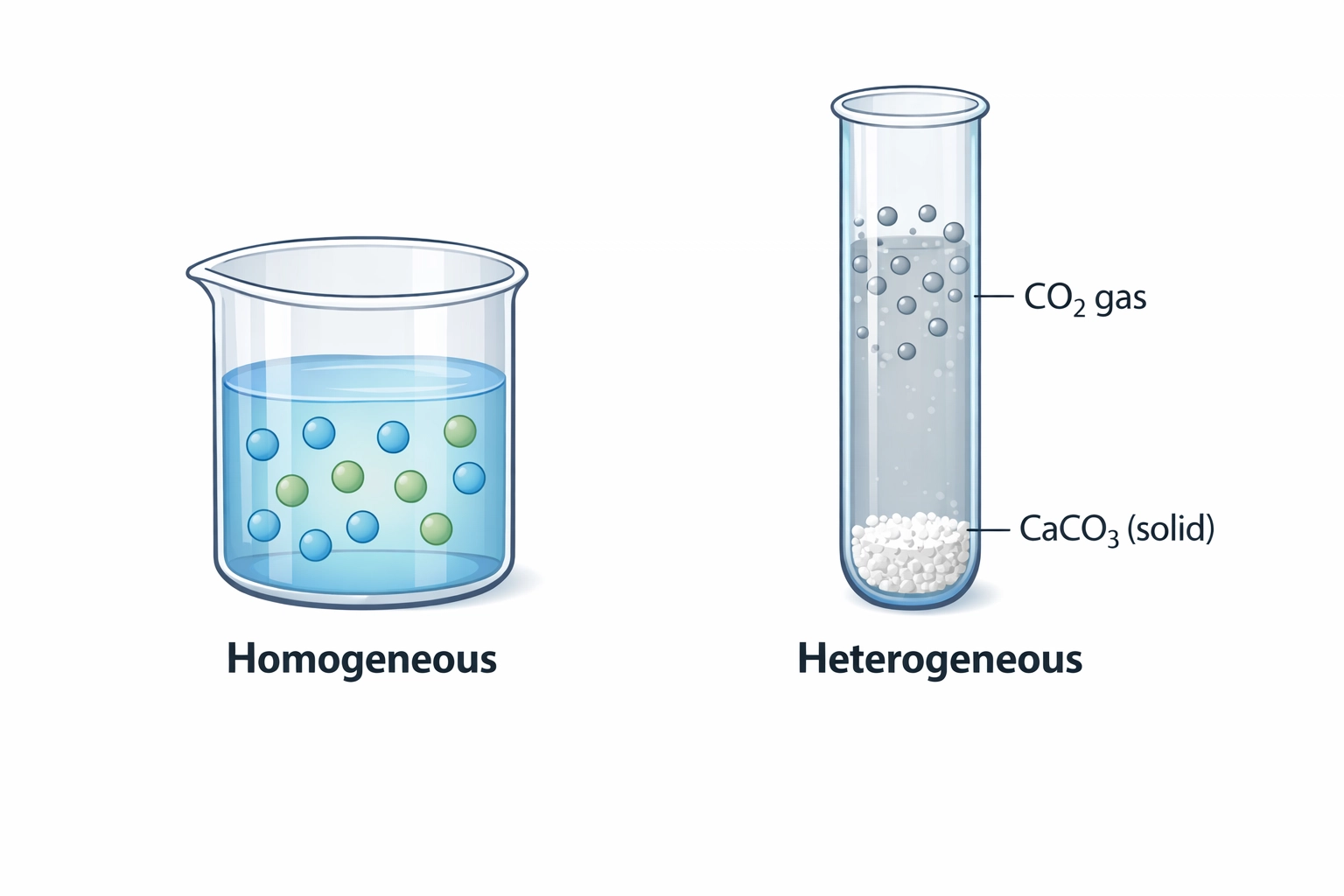
Homogeneous vs. Heterogeneous Systems
This is a specific Edexcel requirement you need to nail.
- Homogeneous: Everything is in the same phase (e.g., all gases or all aqueous). You include everything in the Kc expression.
- Heterogeneous: The substances are in different phases (e.g., a solid reacting with a gas).
The Golden Rule: In heterogeneous equilibria, we ignore solids and pure liquids. We only include gases (g) and aqueous (aq) substances. This is because the concentration of a solid or a pure liquid is considered constant and is essentially “built into” the Kc value.
Example:
CaCO3(s) ⇌ CaO(s) + CO2(g)
Because CaCO3 and CaO are solids, the Kc expression is simply:
Kc = [CO2]
Practical Skills: Seeing Equilibrium in Action
In your Edexcel practicals, you’ll likely see two famous experiments that demonstrate these shifts beautifully.
1. The Iron(III)-Thiocyanate Test
This is a classic “blood-red” reaction.
Fe3+(aq) + SCN-(aq) ⇌ [Fe(SCN)]2+(aq)
(Yellow) + (Colourless) ⇌ (Blood Red)
If you add more Fe3+ (by adding iron(III) nitrate), the solution turns a much deeper red. Why? You’ve increased the concentration of a reactant, so the equilibrium shifts right to produce more red complex. If you add something that reacts with the Fe3+ and removes it, the solution turns back towards yellow.
2. The Cobalt Complex Temperature Test
This one is great for visualizing temperature shifts.
[Co(H2O)6]2+(aq) + 4Cl-(aq) ⇌ [CoCl4]2-(aq) + 6H2O(l)
(Pink) + (Colourless) ⇌ (Blue) + (Colourless)
This reaction is endothermic in the forward direction.
- If you put a tube of this mixture in hot water, it turns blue (shifting right to absorb the heat).
- If you put it in ice water, it turns pink (shifting left to release heat).

Summary Checklist for the Exam
Before you sit your Topic 10 test, make sure you can:
- [ ] Define dynamic equilibrium using the “rates” and “concentrations” keywords.
- [ ] State Le Chatelier’s Principle accurately.
- [ ] Predict shifts for concentration, pressure, and temperature.
- [ ] Explain why catalysts don’t affect the position of equilibrium (they increase the rate of both forward and backward reactions equally).
- [ ] Write Kc expressions, making sure to leave out solids and liquids in heterogeneous systems.
- [ ] Discuss the compromise between yield and rate in industrial processes like the Haber Process.
Equilibrium might seem like a lot of rules to remember, but it all boils down to the system trying to stay balanced. Master the logic of Le Chatelier and the structure of Kc, and you’ll find this topic is one of the most predictable (and high-scoring!) parts of your A Level Chemistry course.
Happy revising! If you need more specific practice questions on calculating Kc units or finding equilibrium concentrations, check out my other deep-dive guides:
Edexcel A Level Chemistry Topic 1: Atomic Structure and The Periodic Table
Edexcel A Level Chemistry Topic 2: Bonding and Structure
Edexcel A Level Chemistry Topic 3: Redox I
Edexcel A Level Chemistry Topic 4: Inorganic Chemistry and the Periodic Table
Edexcel A Level Chemistry Topic 5: Formulae, Equations and Amounts of Substance
