
Edexcel A Level Chemistry Topic 2: Bonding and Structure
Topic 2 is where chemistry really starts to click. Once you understand how atoms bond together and how that bonding affects properties, you’ll find yourself predicting melting points and explaining why ice floats like a pro.
Let’s break it all down.
The Three Types of Bonding
Everything in chemistry comes down to electrons. How atoms share, transfer, or pool their electrons determines everything about a substance’s properties.
Ionic Bonding
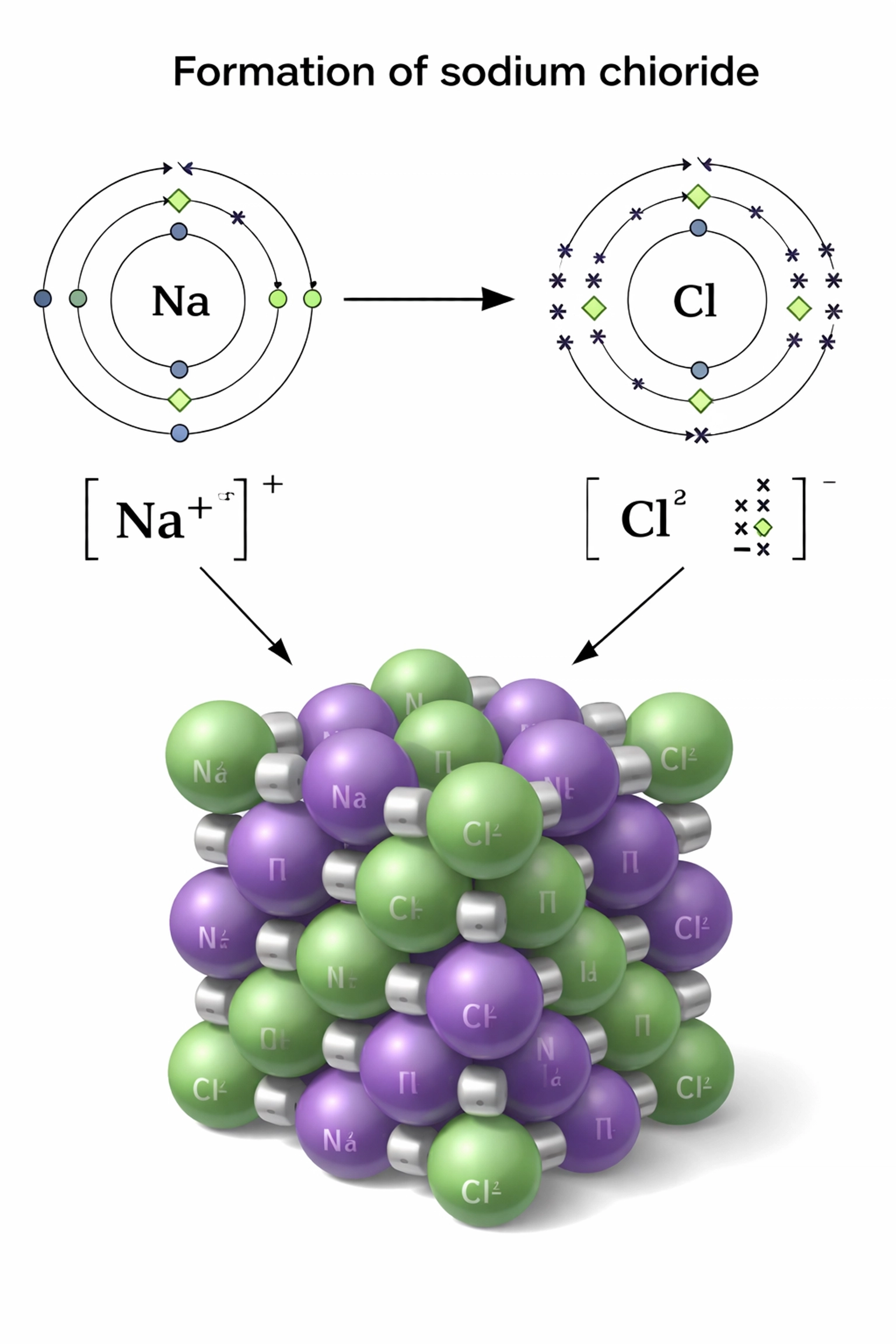
Ionic bonding happens between metals and non-metals. The metal loses electrons to form a positive ion (cation), while the non-metal gains electrons to form a negative ion (anion). These oppositely charged ions are then held together by strong electrostatic attraction.
Two factors affect the strength of ionic bonding:
- Ionic charge: Higher charges = stronger attraction (Mg²⁺O²⁻ is stronger than Na⁺Cl⁻)
- Ionic radius: Smaller ions = stronger attraction (ions are closer together)
When drawing dot-and-cross diagrams for ions, remember to show the electron transfer clearly and include the charge on each ion.
Evidence for ions: When ionic compounds are melted or dissolved in water, they conduct electricity. This proves that mobile charged particles (ions) exist and can carry current.
Covalent Bonding
Covalent bonding occurs between non-metals. Instead of transferring electrons, atoms share them. A covalent bond is the strong electrostatic attraction between two nuclei and the shared pair of electrons between them.
You can have:
- Single bonds (one shared pair)
- Double bonds (two shared pairs)
- Triple bonds (three shared pairs)
Here’s the key relationship: as the number of shared pairs increases, the bond gets shorter and stronger. A C≡C triple bond is shorter and stronger than a C=C double bond, which is shorter and stronger than a C-C single bond.
Dative Covalent (Coordinate) Bonding
Sometimes, one atom provides both electrons in a shared pair. This is called dative covalent or coordinate bonding.
Two examples you must know:
- Al₂Cl₆: Aluminium chloride forms a dimer where a lone pair on chlorine forms a dative bond to an electron-deficient aluminium
- NH₄⁺: When ammonia accepts a proton (H⁺), the nitrogen’s lone pair forms a dative bond to the hydrogen
Once formed, a dative bond is identical to any other covalent bond.
Metallic Bonding
In metals, atoms lose their outer electrons, creating a lattice of positive ions surrounded by a “sea” of delocalised electrons. Metallic bonding is the strong electrostatic attraction between these metal ions and the delocalised electrons.
This explains why metals conduct electricity (mobile electrons), are malleable (layers can slide), and have high melting points (strong bonding throughout).
Shapes of Molecules: VSEPR Theory
Electron-pair repulsion theory (sometimes called VSEPR) predicts molecular shapes. The basic principle: electron pairs around a central atom repel each other and arrange themselves to be as far apart as possible.
Important: Lone pairs repel more than bonding pairs, so they push bonding pairs closer together and reduce bond angles.
Here are the shapes you need to know:
| Electron Pairs | Bonding Pairs | Lone Pairs | Shape | Bond Angle | Example |
|---|---|---|---|---|---|
| 2 | 2 | 0 | Linear | 180° | BeCl₂, CO₂ |
| 3 | 3 | 0 | Trigonal planar | 120° | BCl₃ |
| 4 | 4 | 0 | Tetrahedral | 109.5° | CH₄, NH₄⁺ |
| 4 | 3 | 1 | Trigonal pyramidal | 107° | NH₃ |
| 4 | 2 | 2 | Bent/Angular | 104.5° | H₂O |
| 5 | 5 | 0 | Trigonal bipyramidal | 90° & 120° | PCl₅ |
| 6 | 6 | 0 | Octahedral | 90° | SF₆ |
Electronegativity and Polarity
Electronegativity is the ability of an atom to attract the bonding electrons in a covalent bond. Fluorine is the most electronegative element.
When two atoms with different electronegativities bond, the electrons are pulled towards the more electronegative atom, creating a polar bond with partial charges (δ+ and δ-).
Here’s where it gets interesting: ionic and covalent bonding exist on a continuum. A large electronegativity difference gives ionic character; a small difference gives covalent character.
Polar bonds vs polar molecules: Just because a molecule contains polar bonds doesn’t mean the molecule itself is polar. If the polar bonds are arranged symmetrically, they cancel out.
- CO₂ has two polar C=O bonds, but it’s linear and symmetrical, so it’s non-polar
- H₂O has two polar O-H bonds, but it’s bent, so the dipoles don’t cancel and it’s polar
Intermolecular Forces
These are the forces between molecules, much weaker than covalent bonds but absolutely crucial for understanding physical properties.
London (Dispersion) Forces
Present in ALL molecules. These arise from temporary dipoles caused by random electron movement. The temporary dipole induces a dipole in neighbouring molecules, creating attraction.
Key point: Larger molecules have more electrons, so London forces are stronger. This is why boiling points of alkanes increase with chain length.
Permanent Dipole-Dipole Forces
Occur between polar molecules. The δ+ end of one molecule attracts the δ- end of another.
Hydrogen Bonding
The strongest type of intermolecular force. It occurs when hydrogen is bonded to nitrogen, oxygen, or fluorine (the most electronegative elements with lone pairs).
The δ+ hydrogen on one molecule is attracted to a lone pair on the N, O, or F of another molecule.
Examples: H₂O, NH₃, HF, alcohols
The Anomalous Properties of Water
Hydrogen bonding makes water behave unusually:
- Unusually high melting and boiling points: Water should be a gas at room temperature based on its molecular mass, but extensive hydrogen bonding keeps it liquid.
- Ice is less dense than liquid water: When water freezes, hydrogen bonds hold molecules in an open lattice structure with lots of empty space. This is why ice floats, and why fish survive in frozen ponds!
Boiling Point Trends
Understanding intermolecular forces lets you predict boiling points:
Alkanes:
- Longer chains = more electrons = stronger London forces = higher boiling point
- Branched chains have lower boiling points than straight chains (less surface contact between molecules)
Alcohols vs Alkanes:
Alcohols have much higher boiling points than alkanes with similar numbers of electrons because alcohols can form hydrogen bonds.
Hydrogen Halides (HF to HI):
HCl, HBr, and HI follow the expected trend (boiling point increases with more electrons). But HF has an anomalously high boiling point due to hydrogen bonding.
Solubility: Like Dissolves Like
The golden rule: substances dissolve in solvents with similar intermolecular forces.
- Ionic compounds in water: Water molecules surround and hydrate the ions, breaking down the lattice
- Small alcohols in water: Can form hydrogen bonds with water
- Halogenoalkanes in water: Poor solubility, they’re polar but can’t form hydrogen bonds
- Non-polar substances: Dissolve in non-polar solvents (hexane dissolves in hexane)
Structure Types
Now let’s connect bonding to structure:
| Structure Type | Examples | Melting Point | Conductivity | Solubility |
|---|---|---|---|---|
| Giant ionic | NaCl, MgO | High | Molten/dissolved only | Often water-soluble |
| Giant covalent | Diamond, SiO₂, graphite | Very high | None (except graphite) | Insoluble |
| Giant metallic | All metals | High | Solid and liquid | Insoluble |
| Simple molecular | I₂, ice, CO₂ | Low | None | Depends on polarity |
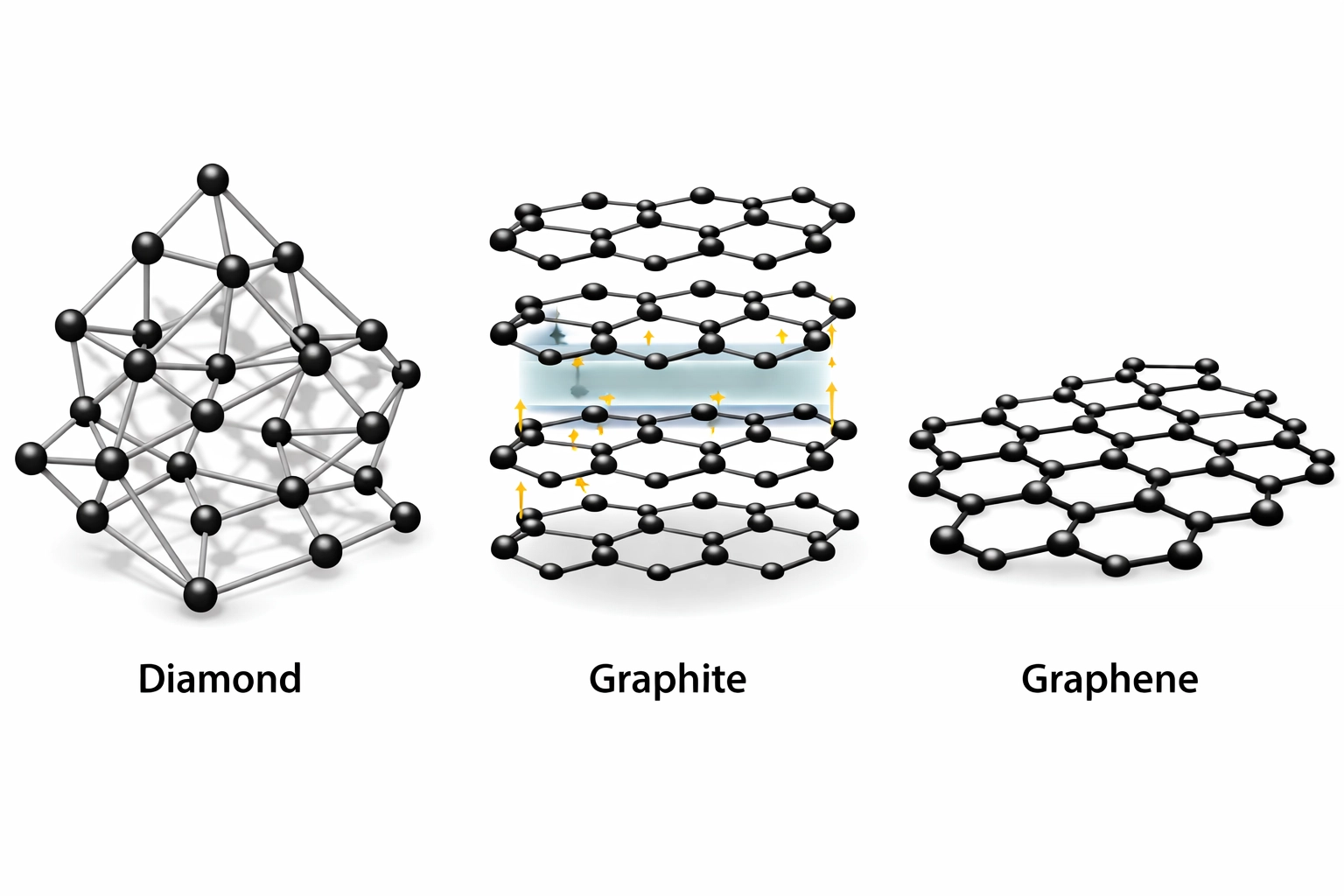
Carbon’s Allotropes
Carbon deserves special attention:
- Diamond: Each carbon bonds to four others tetrahedrally. Very hard, doesn’t conduct electricity.
- Graphite: Layers of hexagonal carbon sheets with delocalised electrons between layers. Conducts electricity, layers slide over each other.
- Graphene: A single layer of graphite. Incredibly strong, conducts electricity.
Predicting Properties from Data
In exams, you’ll often be given data and asked to deduce structure and bonding. Use this checklist:
- High melting point + conducts when molten → Giant ionic
- Very high melting point + doesn’t conduct → Giant covalent
- High melting point + conducts when solid → Metallic (or graphite)
- Low melting point + doesn’t conduct → Simple molecular
Once you’ve identified the structure, you can predict other properties logically.
Final Thoughts
Topic 2 underpins so much of A Level Chemistry. Whether you’re explaining why ethanol mixes with water, why diamond is so hard, or why sodium chloride conducts when molten, you’re applying these same fundamental ideas about bonding and structure.
Master these concepts now, and you’ll find organic chemistry mechanisms, energetics, and even transition metals much easier to understand later on.
