
Edexcel A Level Chemistry Topic 5: Formulae, Equations and Amounts of Substance
Moles and Equations: The Ultimate A Level Chemistry Math Guide!
Let’s be honest: Topic 5 is where A Level Chemistry starts to feel like a maths exam. But here’s the good news: once you nail these calculations, they pop up everywhere else in the course. Master this now, and you’re setting yourself up for success in organic, inorganic, and physical chemistry.
Think of this guide as your calculation cheat sheet. We’ll break down every type of mole calculation, show you exactly how to approach titrations, and make sure you never lose marks to silly unit errors again.
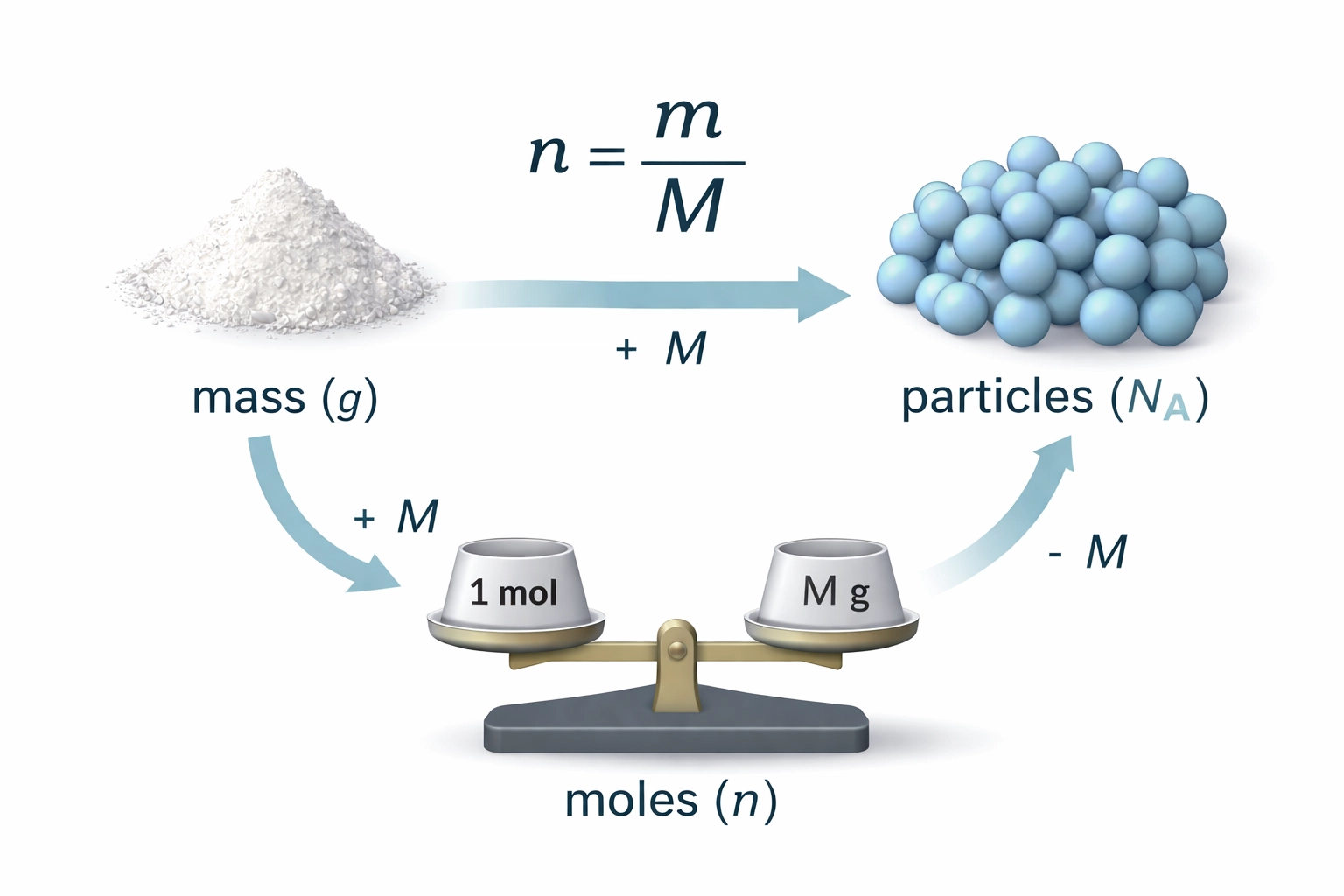
The Mole: Your New Best Friend
A mole is simply a counting unit: like a dozen, but much, much bigger. One mole contains exactly 6.02 × 10²³ particles (atoms, molecules, ions, or electrons). This number is called Avogadro’s constant (L).
Why such a weird number? Because it’s the number of atoms in exactly 12 g of carbon-12. This means the molar mass of any element (in g mol⁻¹) equals its relative atomic mass numerically.
The Holy Trinity of Mole Equations
You’ll use these three equations constantly:
| What you have | Formula | Units |
|---|---|---|
| Mass | n = m / M | g and g mol⁻¹ |
| Solution | n = c × V | mol dm⁻³ and dm³ |
| Gas at RTP | n = V / 24 | dm³ |
Quick conversion reminder: To convert cm³ to dm³, divide by 1000.
Worked Example: Mass to Moles
Calculate the number of moles in 5.30 g of sodium carbonate (Na₂CO₃).
Step 1: Find the molar mass
M = (23 × 2) + 12 + (16 × 3) = 106 g mol⁻¹
Step 2: Apply the formula
n = 5.30 ÷ 106 = 0.0500 mol (3 s.f.)
Empirical vs Molecular Formulae
These two get confused all the time, so let’s clear it up:
- Empirical formula: The simplest whole number ratio of atoms in a compound
- Molecular formula: The actual number of atoms in one molecule
For example, glucose has an empirical formula of CH₂O but a molecular formula of C₆H₁₂O₆.
Finding Empirical Formula (Step-by-Step)
- Write down the mass or percentage of each element
- Divide each by its relative atomic mass (gives moles)
- Divide all values by the smallest number
- If needed, multiply to get whole numbers
Finding Molecular Formula
Once you have the empirical formula:
- Calculate the empirical formula mass
- Divide the molecular mass by the empirical formula mass
- Multiply the empirical formula by this number
Exam tip: If they give you percentage composition, just treat the percentages as masses (assume you have 100 g of the compound).
The Ideal Gas Equation: pV = nRT
This equation lets you find moles of a gas when you’re not at room temperature and pressure. It’s particularly useful for calculating molecular formulae of volatile liquids.
pV = nRT
Where:
- p = pressure in Pa (not kPa!)
- V = volume in m³ (not dm³ or cm³!)
- n = moles
- R = 8.31 J K⁻¹ mol⁻¹
- T = temperature in K (add 273 to °C)
Unit Conversion Cheat Sheet
| From | To | Operation |
|---|---|---|
| kPa | Pa | × 1000 |
| dm³ | m³ | ÷ 1000 |
| cm³ | m³ | ÷ 1,000,000 |
| °C | K | + 273 |
Common mistake alert: The most lost marks in gas calculations come from unit errors. Always convert before you substitute into the equation.
Molar Volume and Core Practical 1
At room temperature and pressure (RTP), one mole of any gas occupies 24.0 dm³ (or 24,000 cm³).
This gives us a beautifully simple equation:
n = V / 24 (when V is in dm³ at RTP)
Core Practical 1: Measuring Molar Volume
In this practical, you’ll react a known mass of a reactive metal (like magnesium) with excess acid, collect the hydrogen gas produced, and use your results to calculate the molar volume.
Key points for write-ups:
- Use a gas syringe or collect over water
- Ensure the gas reaches room temperature before measuring
- Calculate theoretical moles from the mass of metal used
- Compare your experimental volume to the expected 24 dm³ mol⁻¹

Stoichiometry: The Art of Balancing
Every calculation involving chemical equations follows the same three-step process:
- Calculate moles of what you know
- Use the mole ratio from the balanced equation
- Convert to what you need (mass, volume, concentration)
Worked Example: Reacting Masses
What mass of carbon dioxide forms when 25.0 g of calcium carbonate decomposes completely?
Equation: CaCO₃(s) → CaO(s) + CO₂(g)
Step 1: Moles of CaCO₃ = 25.0 ÷ 100 = 0.250 mol
Step 2: From the equation, 1 mol CaCO₃ produces 1 mol CO₂
So moles of CO₂ = 0.250 mol
Step 3: Mass of CO₂ = 0.250 × 44.0 = 11.0 g
Pro tip: Always write the mole ratio from the equation above your working. Examiners love to see this, and it helps you avoid errors.
Titrations: Core Practicals 2 and 3
Titrations are the bread and butter of quantitative chemistry. You’ll need to master both performing them and calculating from the results.
The Titration Calculation
n = c × V
Rearrange as needed:
- c = n / V (to find concentration)
- V = n / c (to find volume)
Core Practical 2: Preparing a Standard Solution
A standard solution has a precisely known concentration. You’ll make one from a solid acid (like potassium hydrogen phthalate) and use it to find the concentration of sodium hydroxide.
Key steps:
- Weigh the solid accurately using a balance (± 0.001 g)
- Dissolve completely in a beaker with distilled water
- Transfer quantitatively to a volumetric flask
- Make up to the graduation mark (meniscus on the line)
Core Practical 3: Finding an Unknown Concentration
Using your standard solution, you’ll titrate against hydrochloric acid of unknown concentration.
Indicator choices:
- Methyl orange: Red in acid, yellow in alkali (use for strong acid + weak base)
- Phenolphthalein: Colourless in acid, pink in alkali (use for weak acid + strong base)
For strong acid + strong base, either works fine.
Percentage Yield vs Atom Economy
These are two different ways to measure how “good” a reaction is: and examiners love asking about both.
Percentage Yield
% yield = (actual yield / theoretical yield) × 100
This tells you how much product you actually made compared to what you should have made. It’s always less than 100% due to:
- Incomplete reactions
- Side reactions
- Loss during transfers and purification
Atom Economy
% atom economy = (molar mass of desired product / sum of molar masses of all products) × 100
This tells you how much of your starting materials end up in the useful product. High atom economy = less waste = better for the environment and industry.
Key distinction: Yield depends on how well you carried out the reaction. Atom economy is fixed by the equation itself: you can’t improve it without changing the reaction.
Errors and Uncertainties
Every measurement has uncertainty. Understanding this helps you evaluate your results and improve your practical technique.
Calculating Percentage Uncertainty
% uncertainty = (uncertainty / measured value) × 100
For example, a 25.00 cm³ pipette with ± 0.06 cm³ uncertainty:
% uncertainty = (0.06 / 25.00) × 100 = 0.24%
Minimising Percentage Error
The golden rule: larger measurements = smaller percentage errors
This is why you should:
- Use a 25 cm³ pipette rather than a 10 cm³ one
- Ensure your titre is a reasonable volume (not 2 cm³)
- Weigh larger masses when possible
Types of Error
| Type | Description | How to reduce |
|---|---|---|
| Random | Unpredictable variations | Repeat and average |
| Systematic | Consistent offset | Calibrate equipment, improve technique |
Quick Reference: Ionic Equations and Observations
For your practical assessments, you need to link equations to observations.
Displacement reactions:
Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s)
Brown deposit forms, blue colour fades
Acid + carbonate:
CO₃²⁻(aq) + 2H⁺(aq) → H₂O(l) + CO₂(g)
Effervescence, gas turns limewater milky
Precipitation:
Ag⁺(aq) + Cl⁻(aq) → AgCl(s)
White precipitate forms
Final Tips for Exam Success
- Show all working: even if you get the wrong answer, you’ll pick up method marks
- Check your units before substituting into any equation
- Use appropriate significant figures: usually 3 s.f. unless the question specifies
- Circle your final answer and include units
- Practise, practise, practise: these calculations become automatic with repetition
Topic 5 might feel like a lot of maths, but every single skill here will come back in later topics. Get comfortable with moles now, and the rest of A Level Chemistry becomes so much more manageable.
