
Edexcel A Level Chemistry Topic 7: Modern Analytical Techniques I
Chemical Detectives: Mastering Mass Spec and IR Spectroscopy! Welcome to the world of chemical forensics! If you’ve ever watched a crime show where they identify mysterious substances in the lab, you’ve seen analytical techniques in action. In Topic 7: Modern Analytical Techniques I, you’ll learn two of the most powerful tools in a chemist’s arsenal: mass spectrometry and infrared spectroscopy. Think of them as your magnifying glass and fingerprint kit for molecules.
These techniques aren’t just for catching criminals (though they do help with drug testing in sport!). They’re how chemists identify unknown compounds, check the purity of medicines, and solve molecular mysteries every single day.
Mass Spectrometry: The Molecular Weighing Scale
Finding the M+ Peak (Your First Clue)
Mass spectrometry works by ionising molecules and then measuring their mass-to-charge ratio (m/z). The beauty of this technique? It tells you exactly how heavy your molecule is.
The molecular ion peak (M+ peak) is the tallest peak with the highest m/z value on the far right of the spectrum. This peak represents your intact molecule that’s lost just one electron. The m/z value of this peak = the relative molecular mass (Mr) of your compound.
Let’s say you’re analysing an unknown alcohol and you spot an M+ peak at m/z = 46. Straight away, you know your molecule has a mass of 46. That’s ethanol (C₂H₅OH), because (2 × 12) + (6 × 1) + 16 = 46.
Top tip for exams: Always look for the M+ peak first. It’s your molecular weight. Sometimes there’s a tiny M+1 peak next to it (due to ¹³C isotopes) – ignore that for now and focus on the main M+ peak.
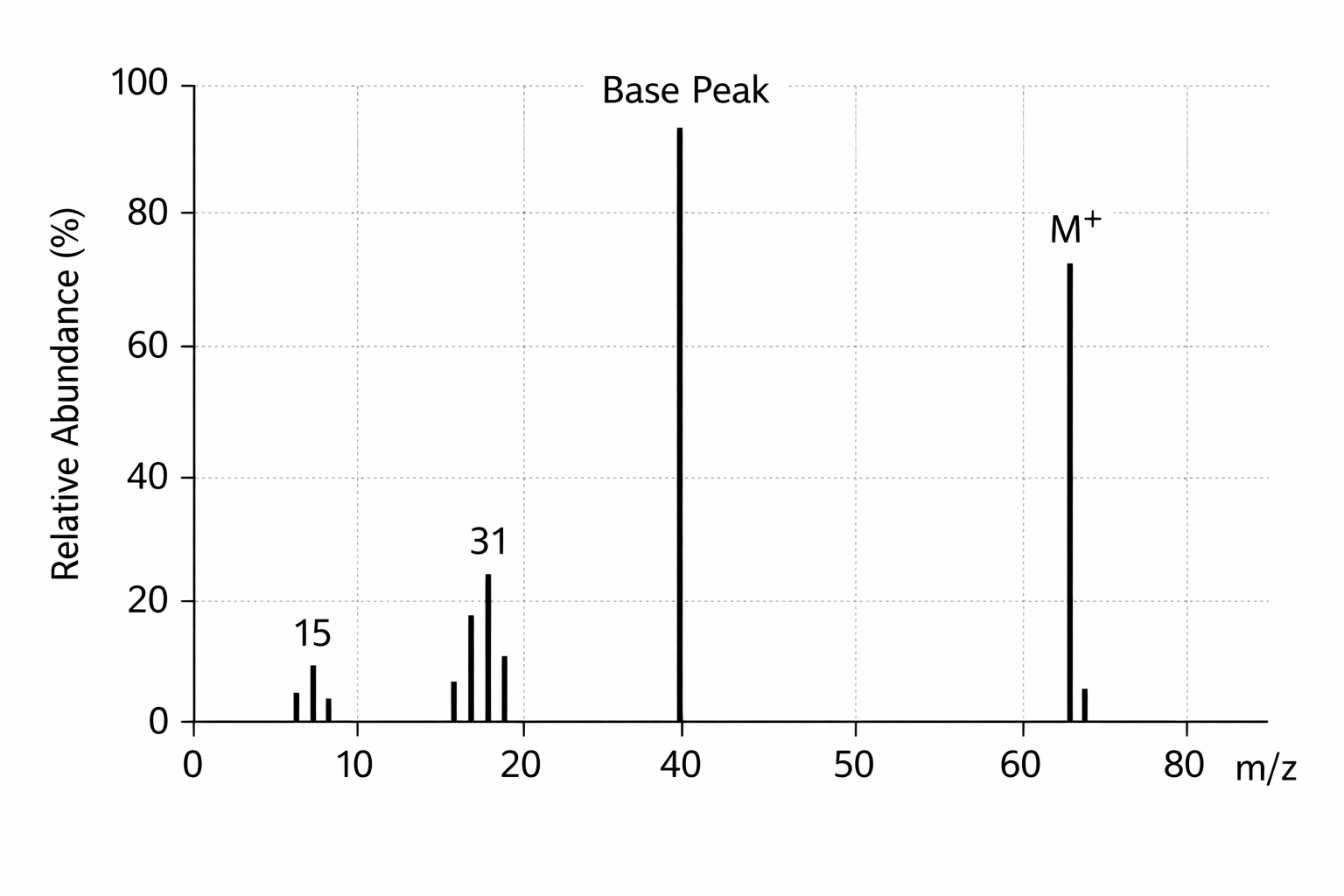
Fragmentation Patterns: The Chemical Jigsaw Puzzle
Here’s where mass spectrometry gets really interesting. When molecules are ionised, they don’t just sit there politely – they break apart into smaller fragments. Each fragment produces its own peak, creating a unique “fingerprint” for that molecule.
These fragments tell you what pieces make up your molecule. It’s like dropping a vase and working out its original shape from the pieces.
Common fragments you’ll see:
- m/z = 15: CH₃⁺ (methyl group)
- m/z = 29: CHO⁺ or C₂H₅⁺ (aldehyde group or ethyl group)
- m/z = 31: CH₂OH⁺ (from alcohols)
- m/z = 43: CH₃CO⁺ (from ketones – this is the acyl cation)
- m/z = 45: COOH⁺ (from carboxylic acids)
How to approach fragmentation in the exam:
- Find the M+ peak (that’s your Mr)
- Look at the other peaks and subtract their m/z from the M+ value
- Work out what fragment was lost
- Use this to deduce the structure
Example: You’ve got an M+ peak at 74 and another significant peak at 45.
- 74 – 45 = 29 (you’ve lost CHO or C₂H₅)
- If you’ve lost 29 and you’re left with 45, you might have lost C₂H₅ from something like C₂H₅COOH (propanoic acid)
- The peak at 45 could be COOH⁺, which confirms the carboxylic acid group
The golden rule: Don’t memorise every possible fragment. Instead, understand that molecules break at weak points (usually next to functional groups or branching points) and learn to recognise the common fragments listed above.
Infrared Spectroscopy: Finding the Functional Groups
If mass spectrometry tells you the molecular weight and gives clues about fragments, infrared (IR) spectroscopy reveals what functional groups are present. It’s like checking the DNA of a molecule.
How IR Spectroscopy Works
Different bonds absorb infrared radiation at specific frequencies (measured in wavenumbers, cm⁻¹). When you shine IR radiation through a sample, certain frequencies get absorbed as bonds vibrate. The resulting spectrum shows “dips” (absorptions) at characteristic wavenumbers.
You don’t need to know the physics – just learn to read the spectrum like a map.
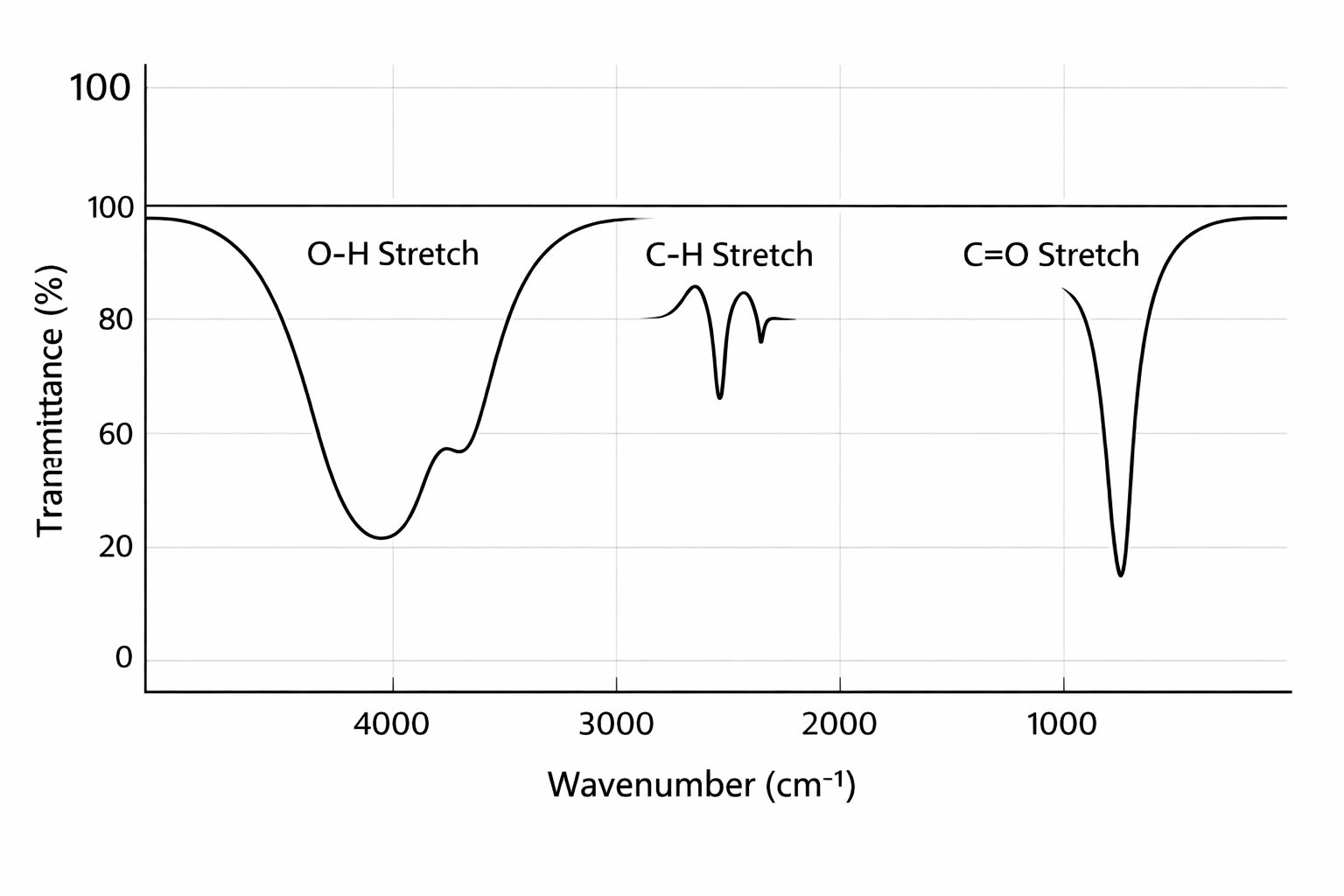
The Key Absorptions You Must Know
Here’s your IR cheat sheet for the Edexcel spec. These are the functional groups you need to identify:
1. O–H in alcohols (3230-3550 cm⁻¹)
- Broad absorption
- Looks like a smooth, wide dip
- Example: If you see this in your spectrum, you’ve got an alcohol (-OH group)
2. O–H in carboxylic acids (2500-3300 cm⁻¹)
- Very broad absorption – much wider than alcohol O-H
- Often looks “stretched out” and messy
- This overlaps with C-H absorptions, creating a distinctive “plateau” shape
- Must also see a C=O peak around 1680-1750 cm⁻¹ to confirm it’s a carboxylic acid
3. C=O in aldehydes and ketones (1680-1750 cm⁻¹)
- Sharp, strong absorption
- One of the most distinctive peaks
- If you also see C-H aldehyde peaks (two small spikes around 2720 and 2820 cm⁻¹), it’s an aldehyde
- If you don’t see those aldehyde C-H peaks, it’s a ketone
4. C=C in alkenes (1620-1680 cm⁻¹)
- Medium strength, sharp peak
- Not always super obvious (can be weak if the alkene is symmetrical)
- Look for =C-H stretches around 3000-3100 cm⁻¹ to support this
5. C–H in alkanes, alkenes, and aldehydes (2850-3100 cm⁻¹)
- Always present in organic molecules (because nearly everything has C-H bonds)
- Alkane C-H: 2850-3000 cm⁻¹
- Alkene =C-H: 3000-3100 cm⁻¹ (just above 3000)
- Aldehyde C-H: Two distinctive peaks at 2720 and 2820 cm⁻¹
6. N–H in amines (3300-3500 cm⁻¹)
- Sharp peak(s)
- Primary amines show two peaks (NH₂ has two N-H bonds)
- Secondary amines show one peak (only one N-H bond)
Reading an IR Spectrum in the Exam
Step 1: Look for the “big three” first:
- Is there a broad O-H? (alcohol or carboxylic acid)
- Is there a sharp C=O? (carbonyl group)
- Is there a C=C? (alkene)
Step 2: Narrow it down:
- Broad O-H + sharp C=O = carboxylic acid
- Broad O-H alone = alcohol
- Sharp C=O + aldehyde C-H peaks = aldehyde
- Sharp C=O without aldehyde peaks = ketone
Step 3: Check for extras:
- Look for N-H if you suspect an amine
- Check the alkene region if you need to identify C=C
Exam tip: You’ll often be given a “window” of the spectrum (maybe just 1600-4000 cm⁻¹) and asked to identify functional groups. Focus on what you can see, not what’s missing from the truncated spectrum.
Combining Your Detective Tools
The real power comes when you use both techniques together. Here’s how:
Scenario: You’re given an unknown compound with the following data:
- Mass spectrum: M+ peak at 60, fragment at 45
- IR spectrum: Broad O-H around 3300 cm⁻¹, no C=O
Your detective work:
- Mr = 60 (from M+ peak)
- Fragment at 45 means you’ve lost 15 (a CH₃ group)
- Broad O-H and no C=O means it’s an alcohol, not a carboxylic acid
- An alcohol with Mr = 60 that loses CH₃ = propan-1-ol or propan-2-ol (C₃H₇OH)
Between propan-1-ol and propan-2-ol? Look back at the mass spectrum. Propan-2-ol has two CH₃ groups that can be lost more easily (fragmentation near branching is favoured), so the m/z = 45 fragment (losing 15) makes sense.
Answer: Propan-2-ol, (CH₃)₂CHOH
Real-World Applications: Why This Matters
These techniques aren’t just for exams. Mass spectrometry combined with IR spectroscopy is used to:
- Detect drugs in athletes’ blood and urine (anti-doping agencies rely on MS for accuracy)
- Identify contaminants in food, water, and medicines
- Test pharmaceutical purity before drugs reach patients
- Analyse crime scene evidence (trace substances, explosives)
- Monitor environmental pollutants
The ability to identify unknown substances quickly and accurately has transformed industries from healthcare to forensics. Understanding these techniques gives you a window into how modern chemistry operates.
Core Practical 7: Analysis of Unknowns
You’ll get hands-on experience with Core Practical 7, where you analyse inorganic and organic unknowns using a combination of techniques. While you won’t be operating a mass spectrometer or IR spectrometer yourself (they’re expensive!), you will:
- Interpret printed mass spectra and IR spectra
- Use test tube reactions (like bromine water for alkenes, Tollens’ reagent for aldehydes)
- Combine multiple pieces of evidence to identify substances
Tips for this practical:
- Make a data table with all your observations – colours, precipitates, gas produced
- Cross-reference chemical tests with spectroscopic data
- Eliminate possibilities systematically (if no C=O in IR, it’s not a carbonyl)
- Show your working – examiners want to see your logical deduction process

Common Exam Mistakes (And How to Avoid Them)
Mistake 1: Confusing alcohol O-H with carboxylic acid O-H
- Fix: Carboxylic acid O-H is much broader (almost a plateau) and you’ll always see a C=O peak as well
Mistake 2: Thinking the tallest peak in a mass spectrum is always M+
- Fix: The tallest peak is the “base peak” (100% abundance). The M+ peak is the peak with the highest m/z value, and it’s often quite small
Mistake 3: Forgetting to subtract fragment peaks from M+ to find what was lost
- Fix: Always show this working: M+ value – fragment value = mass of lost fragment
Mistake 4: Not using all the data provided
- Fix: If you’re given both mass spec AND IR data, you must use both to justify your answer
Mistake 5: Writing vague functional group identifications
- Fix: Don’t just write “carbonyl” – specify aldehyde or ketone using additional evidence (like those aldehyde C-H peaks)
Your Analytical Toolkit
By the end of Topic 7, you’ll be able to:
✓ Identify molecular mass from M+ peaks
✓ Deduce structures from fragmentation patterns
✓ Recognise functional groups from IR absorptions
✓ Distinguish between similar functional groups (aldehydes vs ketones, alcohols vs carboxylic acids)
✓ Combine spectroscopic data to solve molecular mysteries
Think of mass spectrometry as telling you “what’s in the box” and IR spectroscopy as showing you “how the pieces connect.” Together, they’re the ultimate detective duo for identifying organic compounds.
Now grab some practice spectra and start solving those molecular puzzles – your inner chemistry detective is ready! 🔍
