
Edexcel A Level Chemistry Topic 8: Energetics I
Energetics I: The Heat is On! (Mastering Hess’ Law & Calorimetry)
Welcome to Topic 8! If you’ve ever felt like chemistry is just a collection of random facts, Energetics is here to prove you wrong. This is the “accounting” side of chemistry. We aren’t just looking at what molecules are made; we’re looking at the “energy budget.” Where did the heat come from? Where did it go? And most importantly, how do we calculate it without burning the lab down?
In Edexcel Topic 8 (Energetics I), we dive into the world of enthalpy, calorimetry, and the legendary Hess’ Law. Grab your calculator and a cup of tea (which, incidentally, involves energetics to boil): let’s get into it.
1. The Basics: What on Earth is Enthalpy?
Before we start crunching numbers, we need to speak the language. In chemistry, we don’t just talk about “heat.” We talk about Enthalpy (H).
Specifically, we measure the Enthalpy Change (Delta H), which is the heat energy change measured at a constant pressure. Since we usually do experiments in open beakers in a lab, the pressure is constant (atmospheric pressure), so Delta H is our best friend.
The Standard Rules
Examiners love to catch you out on definitions. To make sure everyone’s results are comparable, we use standard conditions:
- Pressure: 100 kPa (about 1 atmosphere).
- Temperature: 298 K (25°C).
- Concentration: 1.0 mol dm⁻³ (for solutions).
When you see that little “Plimsoll” symbol (theta) like this: Delta Htheta, it means the reaction happened under these exact conditions.
The Big Three Definitions
You must memorise these. If you miss out one word (like “one mole”), you lose the mark.
- Standard Enthalpy Change of Formation: The enthalpy change when one mole of a compound is formed from its elements in their standard states under standard conditions.
- Note: The DeltafH of any element in its standard state (like O2(g) or Mg(s)) is always zero.
- Standard Enthalpy Change of Combustion: The enthalpy change when one mole of a substance is completely burned in oxygen under standard conditions.
- Standard Enthalpy Change of Neutralisation: The enthalpy change when solutions of an acid and an alkali react together to form one mole of water under standard conditions.
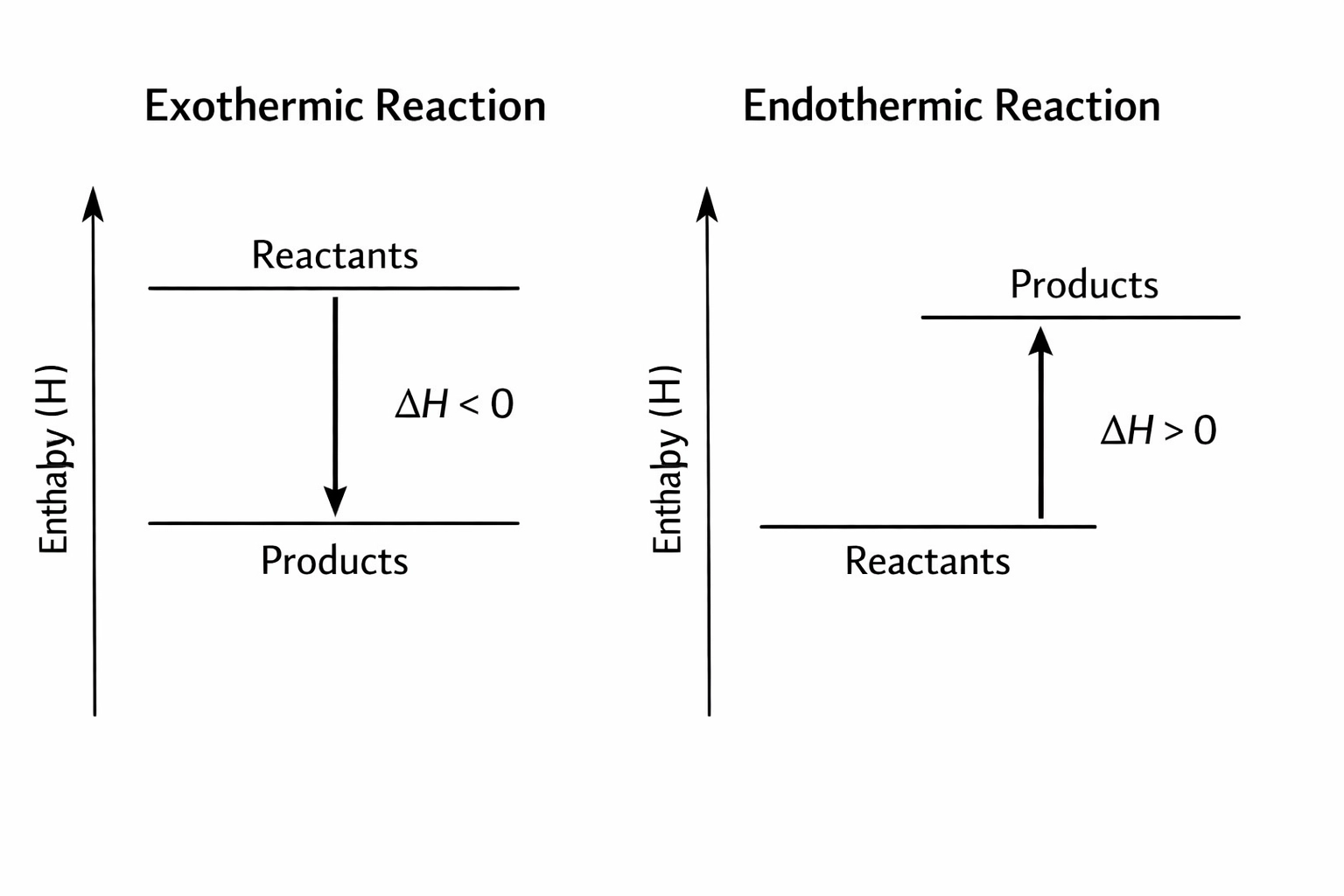
2. Measuring Heat: The Q = mcΔT Equation
How do we actually measure this stuff in a school lab? We use calorimetry.
The logic is simple: if a reaction happens in water and the water gets hotter, the reaction gave off energy (exothermic). If the water gets colder, the reaction sucked energy in (endothermic).
To calculate the energy transferred (Q), we use:
Q = mcΔT
- Q: Energy transferred (in Joules, J).
- m: Mass of the substance being heated (usually the water or the solution). Tip: 1cm³ of water = 1g.
- c: Specific heat capacity (for water, it’s 4.18\ J\ g-1\ K-1).
- ΔT: The change in temperature (Final Temp – Initial Temp).
Converting to Delta H (The Step Everyone Forgets)
Q gives you the total Joules for that specific experiment. But Delta H is usually asked for in kJ mol⁻¹.
To get there:
- Convert Q to kJ (divide by 1,000).
- Calculate the moles (n) of the limiting reactant.
- Use the formula: ΔH = -Q/n
Wait, why the minus sign?
If the temperature goes up, the reaction is exothermic, so Delta H must be negative. If the temperature goes down, the reaction is endothermic, so ΔH must be positive. Don’t lose an easy mark by forgetting the sign!
The Spirit Burner Experiment
A classic exam question involves burning an alcohol in a spirit burner to heat a copper can of water.
Why is the experimental value always lower than the data book value?
- Heat loss to the surroundings (the biggest culprit!).
- Incomplete combustion (you’ll see soot on the bottom of the can).
- Evaporation of the alcohol from the wick.
- Non-standard conditions.
3. Hess’ Law: The Chemist’s GPS
Sometimes, you can’t measure a reaction directly. Maybe it’s too dangerous, too slow, or simply impossible to stop at a specific stage. This is where Hess’ Law saves the day.
Hess’s Law states: The total enthalpy change for a reaction is independent of the route taken.
Think of it like climbing a mountain. Whether you take the steep direct path or the long winding path, the change in your altitude is exactly the same.
Constructing Enthalpy Cycles
There are two main types of cycles you’ll need to master:
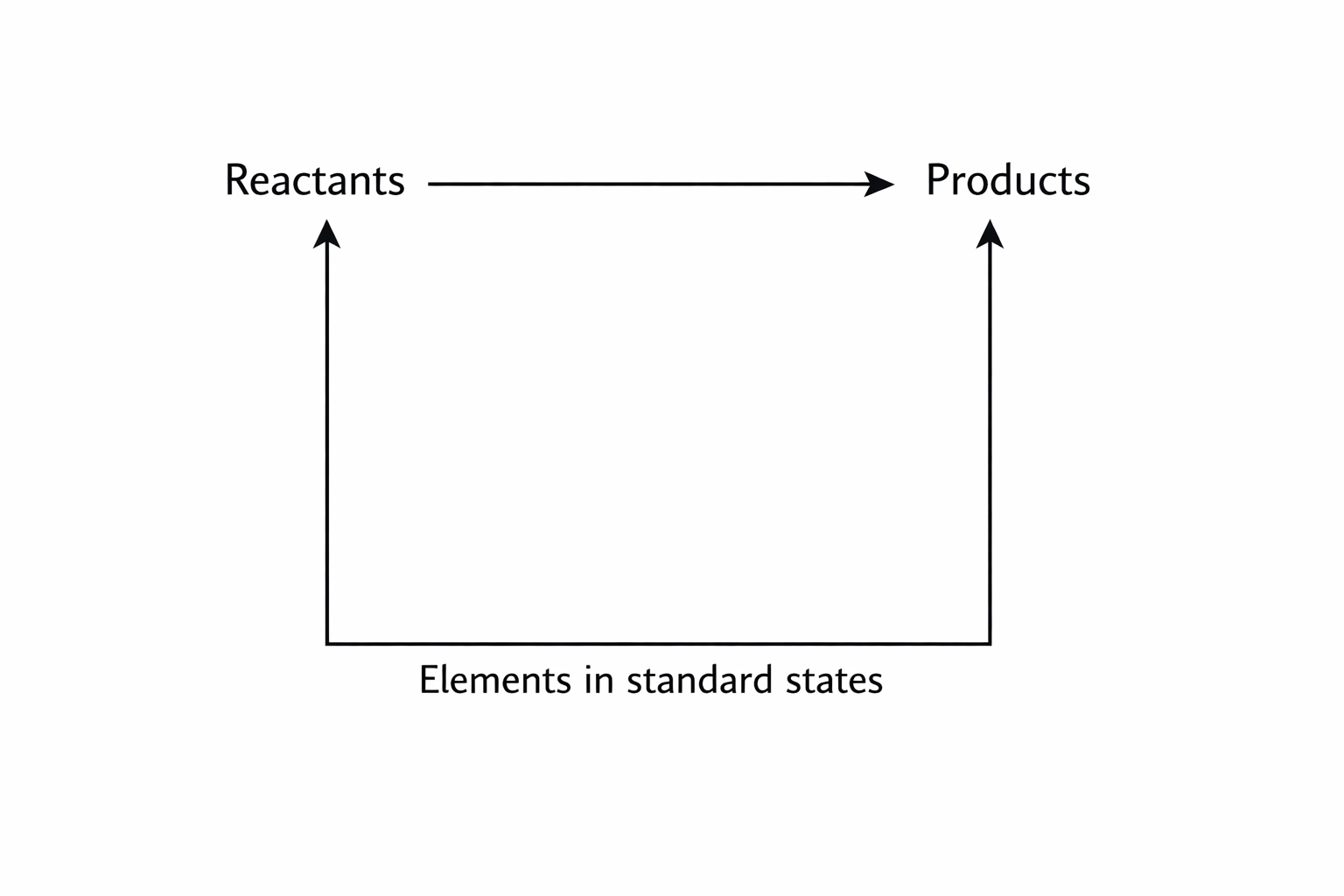
Route A: Using Enthalpies of Formation (ΔfH)
If you are given ΔfH data, your arrows point UP from the elements at the bottom.
ΔH{reaction} = sum ΔfH(products) – sum ΔfH(reactants)
Route B: Using Enthalpies of Combustion (ΔcH)
If you are given ΔcH data, your arrows point DOWN towards the combustion products (CO2 and H2O) at the bottom.
ΔH{reaction} = sum ΔcH(reactants) – sum ΔcH(products)
Pro Tip: Always draw the cycle. It takes 30 seconds and stops you from messing up the signs. Follow the arrows! If you go against an arrow, you must flip the sign of that enthalpy value.
4. Bond Enthalpy: Breaking and Making
If you don’t have formation or combustion data, you can estimate the enthalpy change using bond enthalpies.
- Bond Enthalpy: The energy required to break one mole of a specific bond in a gaseous molecule.
- Mean Bond Enthalpy: The average energy required to break a specific bond, averaged over a wide range of different compounds.
The Golden Rule: “MEXO BENDO”
- Bond Breaking is Endothermic (Requires energy).
- Bond Making is Exothermic (Releases energy).
ΔH = sum (bonds broken) – sum (bonds formed)
Why isn’t it perfect?
You’ll often find that ΔH calculated from bond enthalpies doesn’t match the experimental value perfectly. Why?
- Averages: Mean bond enthalpies are averages. A C-H bond in methane might have a slightly different strength than a C-H bond in an alcohol.
- States: Bond enthalpies are calculated for gases. If your reaction involves liquids or solids, you have to account for the energy needed to change states (latent heat), which bond enthalpies ignore.
5. Core Practical 8: Putting it into Practice
Core Practical 8 usually asks you to determine an enthalpy change that can’t be measured directly. A common example is the transition from anhydrous copper(II) sulfate to hydrated copper(II) sulfate.
You can’t just add exactly enough water to turn a powder into a crystal and measure the temp change easily. Instead, you dissolve both separately in water and use Hess’s Law to find the difference.
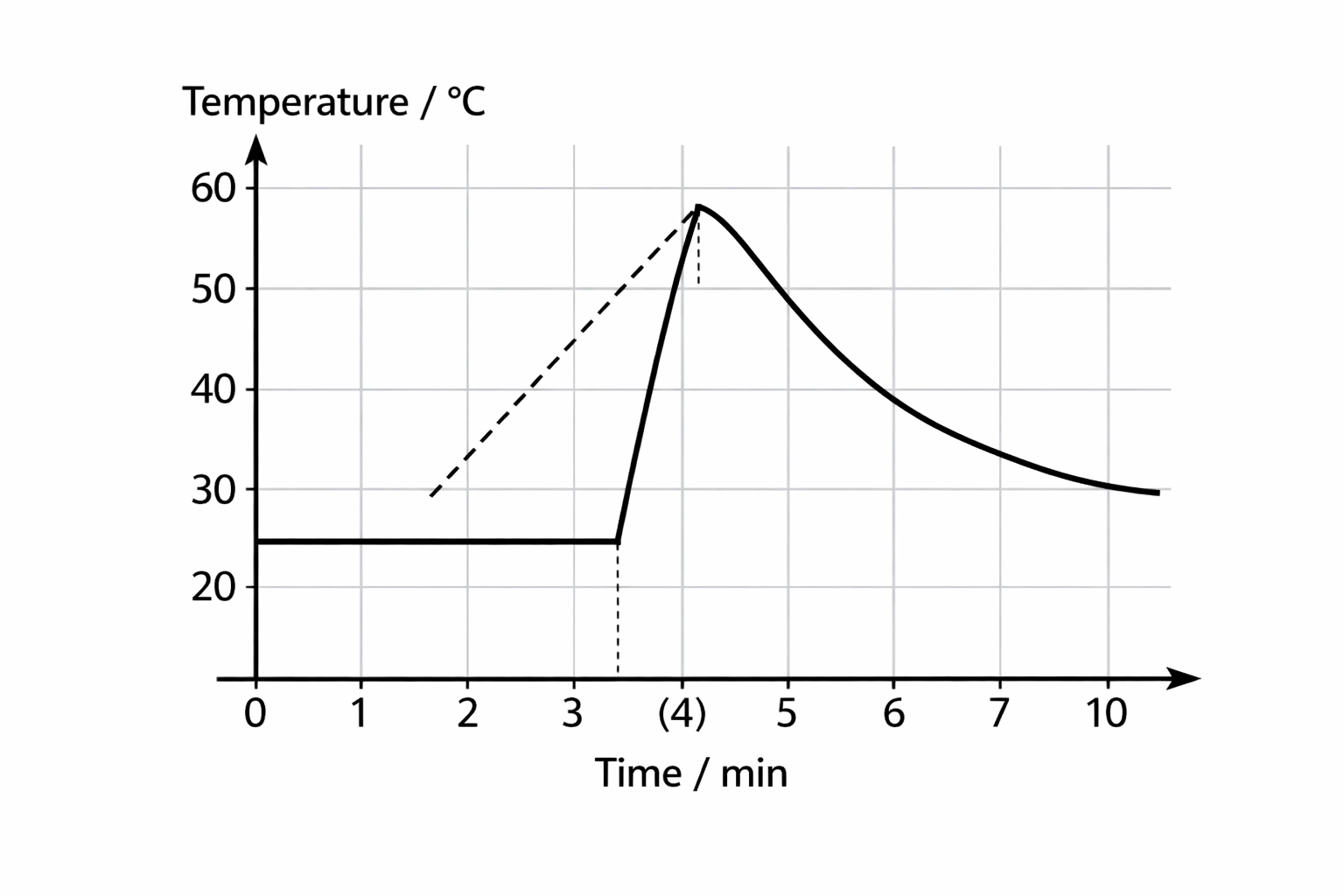
Improving Accuracy: The Graph Method
When you mix two things in a polystyrene cup, the temperature doesn’t just jump instantly; heat is being lost to the room as the reaction happens. To fix this:
- Measure the temperature of the liquid every minute for 3 minutes.
- Add the reactant at the 4th minute (don’t measure).
- Measure the temperature every minute from the 5th to the 10th minute.
- Plot a graph and extrapolate the cooling curve back to the 4th minute. This gives you a more accurate ΔT by accounting for the heat lost during the reaction.
Final Exam Tips for Energetics I
- Watch your units: If Q is in J, remember to turn it into kJ before dividing by moles.
- State Symbols: In enthalpy of formation equations, state symbols are mandatory. H2O(l) is not the same as H2O(g).
- The “Per Mole” trap: If an equation has 2 moles of the substance you’re interested in, you must divide your final ΔH by 2.
- Algebra: When using Hess’s Law, be very careful with double negatives. – (-285) becomes +285.
Energetics might seem like a lot of “plugging numbers into formulas,” but once you see the logic of the cycles, it becomes one of the most satisfying parts of the Edexcel specification. You’re literally calculating the invisible forces that hold the universe together!
Keep practicing those Hess cycles, and remember: Breaking is taking (energy), and making is giving!
Need more help? Check out our worked examples and practice papers in the A Level Chemistry Notes members area. Let’s ace these exams!
