
Edexcel A Level Chemistry Topic 9: Kinetics I
Kinetics I: Speed Dating for Molecules (Mastering Rates & Catalysts)
Welcome to Edexcel A Level Chemistry Topic 9. If Topic 8 was about how much energy is involved in a reaction (Energetics), Topic 9 is about how fast that energy actually does something.
In chemistry, we call this Kinetics. It’s the study of reaction rates. Why do some reactions happen in a fraction of a second (like an explosion), while others take years (like your bike rusting)?
Let’s break down the Edexcel Kinetics I specification into something that won’t make your brain melt.
1. Collision Theory: The Speed Dating Analogy
Think of a chemical reaction like a speed dating event. For a “match” (a reaction) to happen between two molecules, they can’t just exist in the same room. They have to actually meet.
But even meeting isn’t enough. According to Collision Theory, two things must happen for a collision to be “successful”:
- Correct Orientation: The molecules have to hit each other the right way. If the reactive parts of the molecules don’t touch, they’ll just bounce off each other like a bad high-five.
- Sufficient Energy: They have to hit each other hard enough. This minimum amount of energy required for a reaction to occur is called the Activation Energy (Ea).
If they don’t have enough energy, they just bump and part ways. No reaction, no new bonds, no second date.
The Five Factors of Speed
If you want to speed up the dating process (the reaction rate), you have four main levers to pull:
- Concentration: More people in the room = more chances to bump into someone.
- Pressure (for gases): Squeezing the people into a smaller room = even more frequent bumps.
- Surface Area (for solids): Instead of one giant block of marble, use powder. It’s like turning one giant person into a thousand tiny ones: there’s way more “surface” to bump into.
- Temperature: This is the big one. It makes everyone move faster and hit harder.
2. The Maxwell-Boltzmann Distribution
This is a classic exam favorite. The Maxwell-Boltzmann distribution is just a fancy name for a graph that shows the distribution of energies in a gas at a certain temperature.
In any sample of gas, not all molecules are moving at the same speed. Some are slow (low energy), some are incredibly fast (high energy), and most are somewhere in the middle.
Key features of the curve:
- Starts at (0,0): No molecules have zero energy.
- The Peak: This represents the most probable energy of a molecule.
- The Area under the curve: This represents the total number of molecules in the sample.
- The Activation Energy (Ea): We mark this on the x-axis. Only the tiny “tail” of the curve to the right of the Ea line represents molecules with enough energy to actually react.
What happens when you turn up the heat?
When you increase the temperature, two things happen to the curve:
- The peak shifts to the right (the average energy increases).
- The peak gets lower (to keep the total area under the curve the same).
The most important part? A much larger proportion of molecules now sit to the right of the Ea line. Note for the exam: The main reason a higher temperature speeds up a reaction isn’t just because there are more collisions, it’s because a much higher fraction of those collisions are successful.
3. Measuring the Rate (Math Alert!)
How do we actually measure “speed” in chemistry? We look at how fast a reactant disappears or how fast a product appears.
Rate = Change in concentration\Time
Plotting the Data
You’ll often be asked to plot a graph of concentration (or volume of gas) against time.
- The reaction is fastest at the start (steepest gradient) because the concentration of reactants is at its highest.
- The reaction slows down as reactants are used up.
- The curve flattens out when the reaction stops.
The Tangent Trick
To find the rate at a specific time (t), you can’t just read the graph. You have to draw a tangent (a straight line that just touches the curve at that point) and calculate its gradient:
Gradient = Delta y\Delta x
If they ask for the initial rate, draw your tangent at t = 0
4. Catalysts: The Ultimate Shortcut
A catalyst is like a GPS that finds a shortcut to the finish line. It speeds up a reaction without being used up itself.
How? By providing an alternative reaction pathway with a lower activation energy.
Think of it this way: If you’re trying to get to the other side of a mountain, the “uncatalysed” route is climbing over the peak. The “catalysed” route is taking a tunnel through the middle. You end up in the same place, but it takes way less energy to get there.
Reaction Profiles
You need to be able to draw these. The “hump” represents the activation energy. When you add a catalyst, you draw a second, lower hump.
Crucial Point: The overall enthalpy change stays exactly the same. The starting and ending points don’t move; only the height of the hill changes.
5. Industrial Catalysts & The Economy
In the real world (and in Edexcel exams), catalysts aren’t just cool science: they’re worth billions of pounds.
Most industrial catalysts are heterogeneous. This means they are in a different phase (usually a solid) than the reactants (usually gases).
How it works (Surface Chemistry):
- Adsorption: Reactant molecules land on the surface of the solid catalyst and stick to “active sites.”
- Reaction: The bonds in the reactants are weakened, making it easier for them to break and reform into products.
- Desorption: The product molecules break away from the surface, leaving the active site free for the next set of reactants.
Why do we care?
- Lower Temperatures: Because the Ea is lower, we don’t need to blast the furnace as hot. This saves massive amounts of money on fuel.
- Environmental Impact: Less fuel burned = fewer CO2 emissions.
- Better Yield: We can make products faster, making the whole process more profitable.
6. Common Exam Pitfalls (Don’t Lose Marks Here!)
Even if you understand the concepts, examiners love to catch you out on the details. Here are a few things to keep in mind:
- State Symbols: If you’re writing an equation for a reaction involving a catalyst (like the Haber Process), make sure you know the states.
- “Frequent” vs “Successful”: Never just say “there are more collisions.” You must say there are “more frequent collisions” or “more collisions per second.” Even better, say “a greater proportion of collisions are successful.”
- Maxwell-Boltzmann Labels: Make sure you label the axes correctly. Y-axis is “Number of molecules” (or fraction of molecules), and X-axis is “Kinetic Energy.” Don’t swap them!
- Activation Energy Definition: It is the minimum energy required for a collision to result in a reaction. Don’t forget the word “minimum.”
7. Practical Skills: The Core Experiments
For Topic 9, you should be familiar with a few key experiments:
- Disappearing Cross (Sodium Thiosulfate + HCl): You time how long it takes for a precipitate of sulfur to form and obscure a cross drawn on paper. This is a classic way to investigate temperature or concentration.
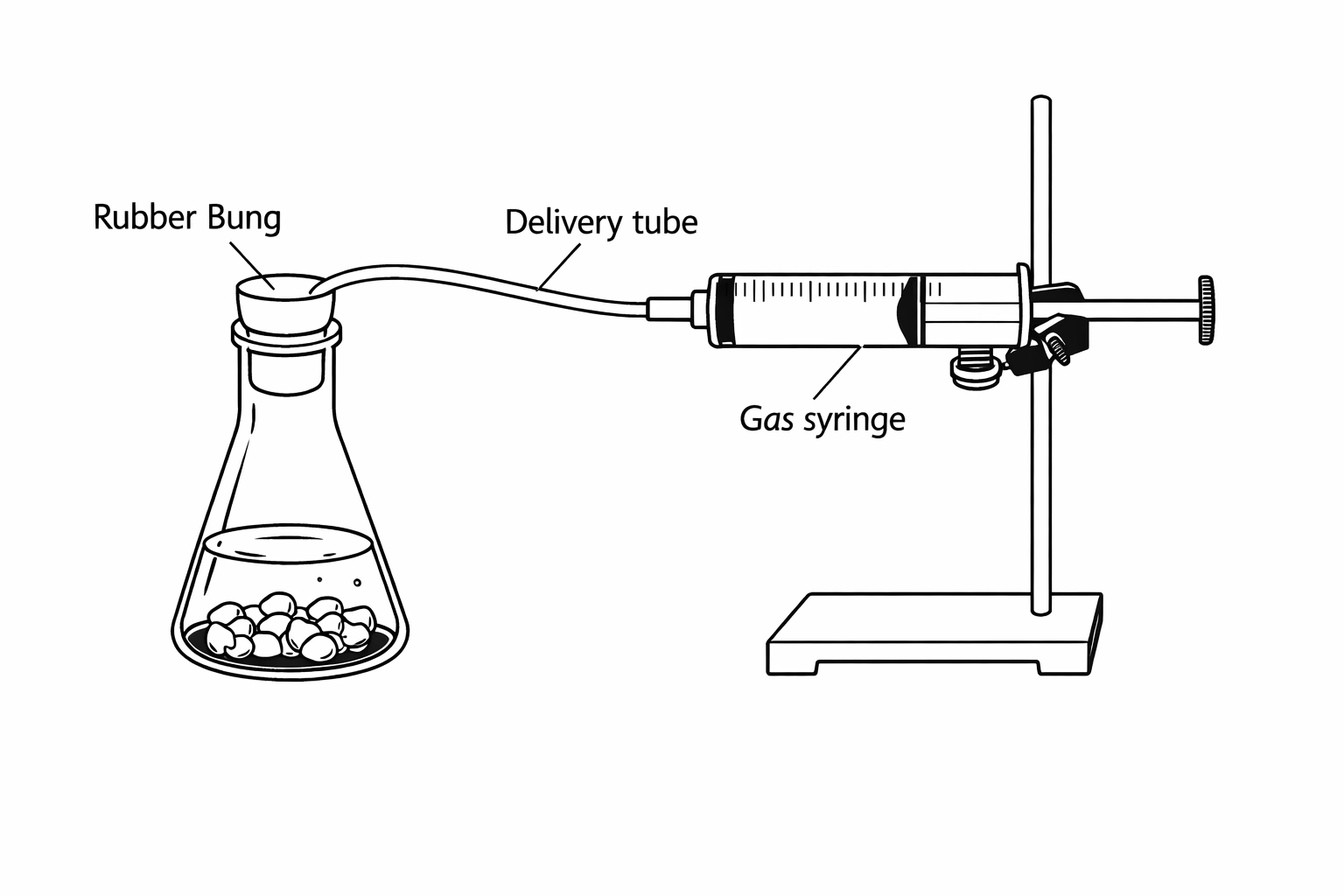
- Gas Collection: Reacting marble chips (CaCO3) with acid and measuring the volume of CO2 produced using a gas syringe. This is great for looking at surface area.
- Catalysis of H2O2: Using Manganese(IV) Oxide to speed up the decomposition of hydrogen peroxide.
When discussing these in exams, always think about errors. Is the gas syringe sticking? Are you losing gas before you can put the bung in? Did you start the stopwatch exactly on time?
Summary Checklist
Before you move on to Topic 10, make sure you can:
- [ ] Explain Collision Theory using the “orientation” and “energy” requirements.
- [ ] Describe how concentration, pressure, and surface area affect frequency.
- [ ] Draw and explain the Maxwell-Boltzmann distribution (and the shift with temperature).
- [ ] Define Activation Energy accurately.
- [ ] Calculate a reaction rate from a graph using a tangent.
- [ ] Draw a reaction profile showing the effect of a catalyst.
- [ ] Explain how a heterogeneous catalyst works (Adsorption/Desorption).
Kinetics I is all about the “why” and the “how fast.” Master the Maxwell-Boltzmann curve and the definition of a catalyst, and you’re already halfway to an A.
Ready to tackle some past paper questions? Go get ’em! 🚀
