
Blog
The ‘Mole Mountain’: Why Chemistry Calculations Trip Up Even the Brightest Students

You’ve aced every theory question on the paper. Your organic mechanisms? Flawless. Your knowledge of trends in the periodic table? Absolutely solid. But then you flip the page and see it: a 4-mark calculation staring back at you. Your stomach drops. The numbers blur together. And suddenly, all that confidence you built up? It evaporates. 🌪️
If this sounds familiar, you’re not alone. I’ve worked with countless bright, hardworking A-Level and IB students who genuinely believe they’re “just not math people.” But here’s the truth I want you to hear: it’s not about being a math person, it’s about having the right framework. And that’s exactly what most students are missing.
The Complexity Jump: When Chemistry Gets Real
Let’s talk about what actually changes between GCSE and A-Level Chemistry calculations. At GCSE, you’re dealing with straightforward, one-step problems. Calculate the number of moles? Sure, divide mass by molar mass, done. Work out concentration? Easy formula, plug in the numbers, move on.
But A-Level? That’s where things get spicy. 🔥
Suddenly, you’re facing multi-step, multi-unit conversion nightmares where everything seems connected to everything else. You need to convert dm³ to cm³, then grams to moles, then apply stoichiometry from a balanced equation, then calculate percentage yield, all in one question. And if you mess up step two? The entire house of cards comes tumbling down.

The real challenge here is what I call “the invisible bridge.” The mole concept is fundamentally abstract, it represents 602 hexillion particles that you cannot directly observe or measure. You’re being asked to build a bridge between what you can see and measure (mass, volume) and what you can’t (individual atoms and molecules). That’s a massive conceptual leap, and it’s not surprising that even the brightest students find themselves stuck halfway across.
The Memorization Trap: Why Formula Triangles Fail You
I see this all the time: students armed with pages of formula triangles, desperately trying to remember which letter goes where. And yes, at GCSE, this might have carried you through. But here’s the uncomfortable truth: memorization without understanding is a trap.
When exam questions become contextual, when they throw you a curveball about an unfamiliar reaction or ask you to work backwards from a product, those memorized triangles become useless. You need to understand why you’re dividing mass by molar mass, not just that you should.
The mole isn’t just some arbitrary unit your teacher invented to torture you. It’s actually the foundation of stoichiometry, the crucial link between chemical equations (which deal in ratios of particles) and real-world quantities (which we measure in grams and litres). Without genuinely understanding this connection, you’re building on sand.
And here’s where students really get stuck: you’re being asked to simultaneously juggle:
- Avogadro’s number (6.02 × 10²³) as a standard counting unit
- Molar mass as the mass of exactly one mole of a substance
- Conversion pathways between mass, moles, volume, concentration, and particle number
- Stoichiometric ratios from balanced equations
That’s a lot of plates to spin. No wonder it feels overwhelming. 🎪
The Solution: The ‘Roadmap’ Approach
Alright, here’s where things get better. I promise. 💚
After years of working with students who were convinced they’d “never get calculations,” I developed what I call The Roadmap Approach. It’s simple, it’s repeatable, and it works for literally every calculation question you’ll encounter.
Step 1: Identify the ‘Destination’
Before you write a single number, ask yourself: What am I actually finding? Circle it. Underline it. Make it crystal clear. Are you calculating mass? Volume? Number of moles? Percentage yield?
This sounds ridiculously simple, but you’d be amazed how many students dive straight into calculations without knowing where they’re heading. It’s like setting off on a road trip without knowing your destination, you might drive for hours and end up nowhere useful.
Step 2: List Your ‘Supplies’
Now look at what the question has given you. What data do you have? Write it all down with units. This is your starting inventory. You might have:
- A mass in grams
- A volume in cm³ or dm³
- A concentration in mol dm⁻³
- A balanced equation with stoichiometric ratios
Pro tip: If units are missing or need converting, flag them now. Converting dm³ to cm³ at the end because you forgot? That’s how marks slip away.
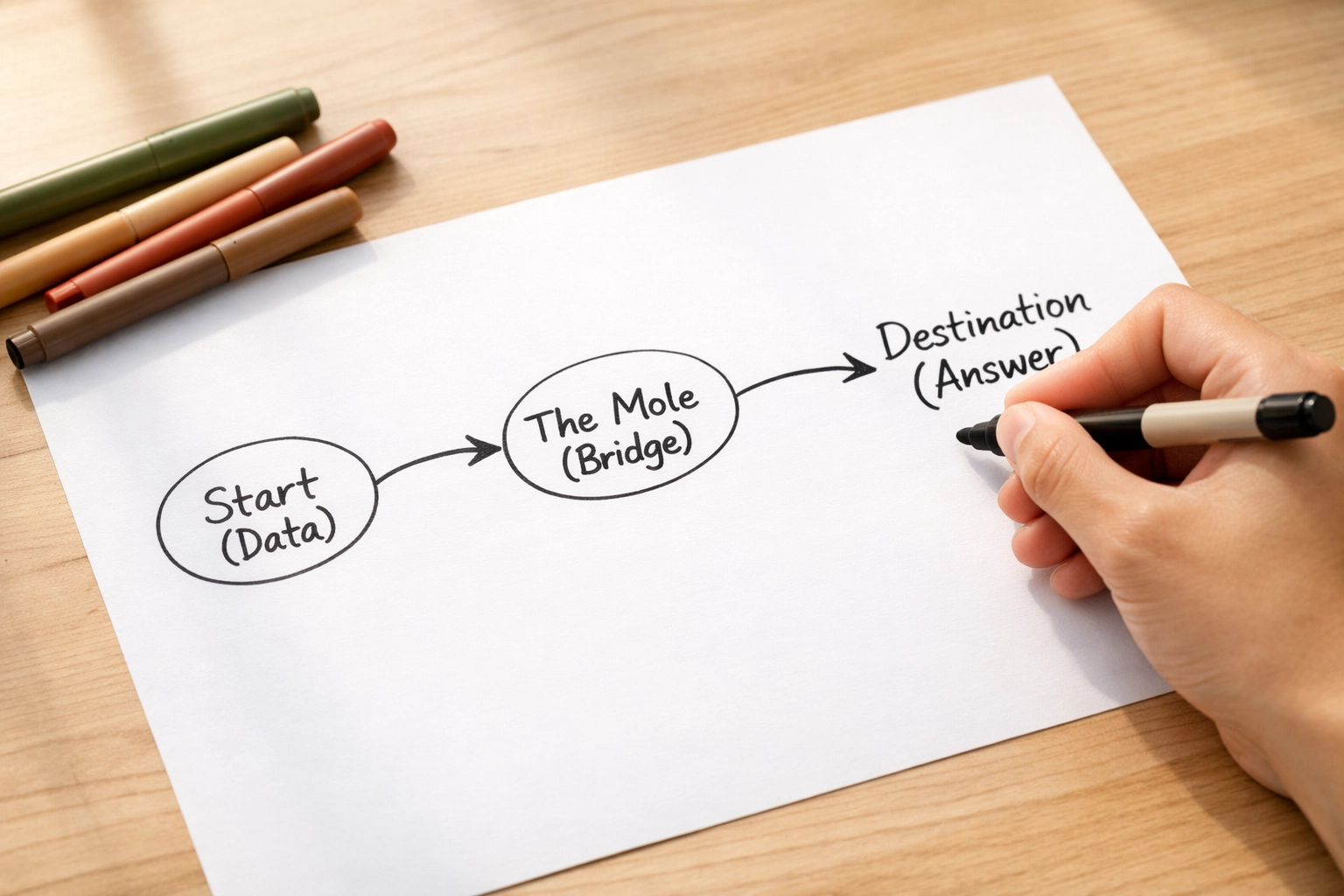
Step 3: The Bridge (The Mole)
Here’s the golden rule that changes everything: Everything goes through the mole.
No matter what you’re converting: mass to particles, volume to mass, concentration to yield: the mole is your bridge. It’s the universal translator of chemistry. Once you’ve got your data in moles, you can convert it to anything else using the relationships you know:
- Moles = mass ÷ molar mass
- Moles = concentration × volume (in dm³)
- Moles = particles ÷ Avogadro’s number
And stoichiometry? That’s just using the ratio from your balanced equation to convert between moles of different substances.
When you approach calculations this way, they stop being a tangled mess and start being a logical sequence of conversions. Step by step. Always passing through the mole.
The Strategic Advantage: Removing Background Anxiety
Let’s talk about something that doesn’t get discussed enough: calculation anxiety bleeds into the rest of your exam.
When you’re terrified of the calculations section, it creates this low-level stress that runs throughout the entire paper. You’re mentally calculating how many marks you’ll lose, how much pressure that puts on your theory questions, whether you can still hit your target grade if you bomb this section.
That’s exhausting. And it affects your performance on questions you should be nailing.
But here’s the flip side: when you master calculations early, everything changes. Suddenly, you walk into that exam knowing that 20-30% of the paper is locked in. Those marks are yours. The background anxiety disappears, replaced by quiet confidence. And that confidence? It shows up in your theory answers, your explanations, your ability to think clearly under pressure.
Mastering calculations isn’t just about those specific marks: it’s about transforming your entire exam experience. 🌟
The Expert Edge: Pattern Recognition Over Memorization
When I work with students as an A Level Chemistry tutor, my focus isn’t on drilling formulas until they stick. Instead, we work on pattern recognition: training your brain to see the structure underlying every calculation, not just a wall of intimidating numbers.
After a few sessions, something beautiful happens: you start spotting the patterns yourself. You see a question about empirical formula and immediately recognize it’s a “mass → moles → ratio” pathway. You see “dm³” and automatically think “multiply by concentration to get moles.” The calculation stops being a puzzle you need to solve from scratch every time and becomes a familiar journey you’ve walked before.
This is particularly valuable for students working with an online chemistry tutor, where we can screen-share calculations, break them down step-by-step in real-time, and build that pattern recognition systematically. Whether you’re studying in London or working with a chemistry tutor Dubai via video call, this approach translates beautifully: because the patterns are universal.

And here’s what I tell every student I work with: your weakest area can become your competitive advantage. The students who struggle with calculations but push through and master them? They often end up outperforming students who found them easy at first but never developed rigorous technique. Because they’ve built an unshakeable foundation.
Your Move: From Ceiling to Springboard
Look, I get it. Calculations feel like the ceiling on your grade right now. That hard limit you keep bumping up against, no matter how much organic chemistry or energetics you master. But they don’t have to be.
With the right framework, calculations become the springboard that launches your grade upward. They become the section where you collect easy marks while other students panic. They become your secret weapon.
If you’re tired of feeling like “not a math person,” if you’re ready to turn your weakest area into your strongest competitive advantage, I’d love to work with you. My sessions are specifically designed to rewire how you approach calculations: not through endless repetition, but through genuine understanding and pattern recognition.
Don’t let calculations be the thing that holds back an otherwise excellent Chemistry grade. You’re smarter than you think. You just need someone to show you the roadmap. 🗺️
Book a session today and let’s turn that Mole Mountain into a hill you climb with confidence.
Oxford-Educated Chemistry Specialist
With over 20 years of teaching experience at some of the UK’s top independent schools, I help ambitious students bridge the gap between hard work and top-tier results. I specialise in GCSE, A Level, and IB Chemistry tuition for students targeting Grade 9s and A*s. Based in the UK but working globally, I provide 1-1 online support for families in South and West London, Dubai, and Hong Kong, ensuring students are perfectly prepared for competitive medical applications and Oxbridge entries.
I’ve helped students achieve top grades from schools such as Alleyn’s, Dulwich College, Tonbridge, Sevenoaks, Brighton College, Wycombe Abbey, Caterham, St Paul’s, Dubai College, Dubai British School and Harrow International School Hong Kong.
Contact me archardchloe@gmail.com to discuss how I can help your child excel in Chemistry.
