
Blog
Why Do Transition Metals Look So Cool? The Real Chemistry Behind Their Colour and Tricks

Ever wondered why copper sulfate crystals sparkle with that gorgeous blue colour, or why iron rust turns that distinctive orange-brown? 🌟 The answer lies in one of chemistry’s most fascinating topics: transition metals and their incredible ability to create a rainbow of colours that would make any artist jealous!
If you’re studying GCSE or A Level Chemistry, understanding why transition metals are colourful isn’t just about acing your exams (though it definitely helps with that too 😉). This knowledge unlocks the secrets behind everything from stained glass windows in ancient cathedrals to the modern paint on your bedroom walls.

Let’s dive into the real chemistry behind these colour-changing superstars and discover why they’re absolutely essential for your chemistry success!
What Makes Transition Metals So Special?
Transition metals are the elements sitting in the middle block of the Periodic Table – think scandium through to zinc in the first row. But here’s what makes them truly special: they have partially filled d-orbitals.
Unlike your typical sodium or chlorine atoms, transition metals have d-electrons that can do some pretty amazing tricks. These electrons are the key players in creating the spectacular colours we see in transition metal compounds.

When you dissolve a transition metal salt in water, something magical happens. The metal ions don’t just float around alone – they get surrounded by water molecules (or other ligands), forming what chemists call complex ions. This is where the colour story really begins!
The Science Behind the Rainbow 🌈
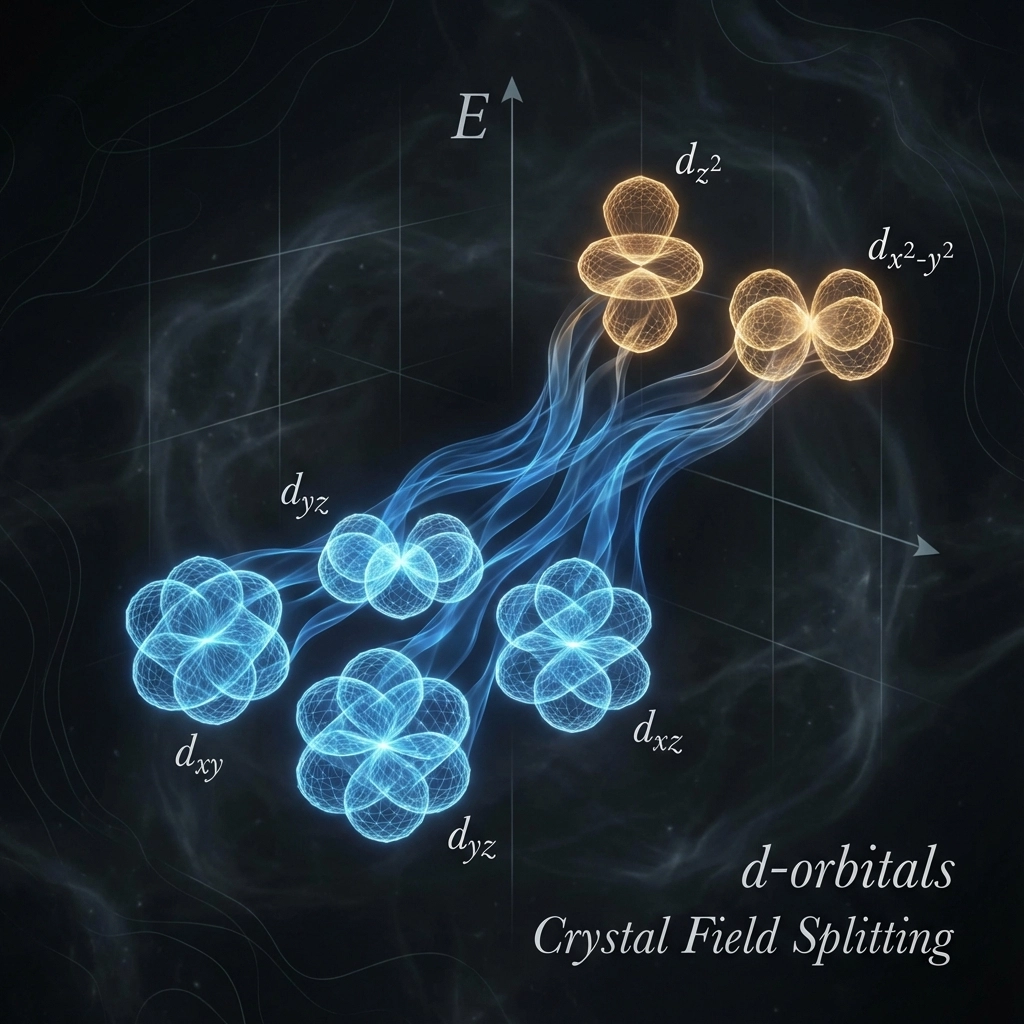
Here’s where it gets really exciting! When transition metal ions form complexes, their five d-orbitals split into different energy levels – a process called crystal field splitting. Think of it like taking five identical shelves and suddenly making some higher and some lower.
In most cases (specifically octahedral complexes), the d-orbitals split into two groups:
- Three lower-energy orbitals (called t₂g)
- Two higher-energy orbitals (called eg)
Now here’s the clever bit: electrons in these d-orbitals can absorb energy and jump from the lower-energy group to the higher-energy group. This is called a d-d transition, and it’s the secret behind transition metal colours!
The energy needed for this jump corresponds to specific wavelengths of visible light. When white light hits your solution, certain colours get absorbed as electrons make these jumps. The colour you actually see is the complementary colour of what’s been absorbed.
Pro tip for exams: If a complex absorbs red light, it appears green. If it absorbs blue light, it appears orange. This complementary colour relationship is crucial for understanding transition metal chemistry! ⭐
What Determines Which Colour You See?
Several factors work together to determine the final colour of a transition metal complex:
The Metal Ion Identity
Different metals have different numbers of d-electrons and different splitting energies. This is why copper(II) compounds typically appear blue, while iron(III) compounds are yellow or brown.
Oxidation State Matters
The oxidation state of the metal ion dramatically affects colour. Compare these common examples:
- Fe²⁺ (iron II): Pale green solutions
- Fe³⁺ (iron III): Yellow to brown solutions
Higher oxidation states pull ligands in more strongly, increasing the crystal field splitting and changing the energy gap between orbitals.
Ligand Type Makes a Difference
Different ligands (the molecules or ions surrounding the metal) create different amounts of splitting:
- Strong-field ligands (like CN⁻ and NH₃) create large splitting, often resulting in yellow, orange, or red complexes
- Weak-field ligands (like H₂O and halides) create smaller splitting, typically producing blue, green, or purple complexes
Geometry Changes Everything
Octahedral, tetrahedral, and square planar complexes split d-orbitals in completely different ways, producing distinct colour patterns.
Transition Metals in Everyday Life
Understanding transition metal colours isn’t just academic – these principles are everywhere around you! 🙌
Stained Glass and Art
Those magnificent medieval cathedral windows? They get their deep blues from cobalt, rich greens from chromium, and brilliant reds from gold nanoparticles. Artists have been using transition metal pigments for centuries because they’re incredibly vibrant and stable.
Industrial Testing and Analysis
In laboratories around the world, chemists use transition metal colours to identify unknown substances. The flame test colours you might do in class are just the beginning – complex colour changes help detect specific ions in water samples, medicines, and industrial products.
Modern Technology
From the catalytic converters in cars (using platinum and rhodium) to the rechargeable batteries in your phone (containing lithium, cobalt, and nickel), transition metals are absolutely essential for modern life.

Your Essential Colour Reference Guide
Here’s a handy table for your revision notes – bookmark this for quick exam reference! 📚
| Metal Ion | Colour in Aqueous Solution | Common Compound |
|---|---|---|
| Cu²⁺ | Bright blue | Copper sulfate |
| Fe²⁺ | Pale green | Iron(II) sulfate |
| Fe³⁺ | Yellow/brown | Iron(III) chloride |
| Cr³⁺ | Green/violet | Chromium chloride |
| Co²⁺ | Pink | Cobalt chloride |
| Ni²⁺ | Green | Nickel sulfate |
| Mn²⁺ | Pale pink | Manganese sulfate |
Why Don’t All Elements Have Coloured Compounds?
This is a favourite exam question, so pay attention! 🎯
Alkali metals (like sodium and potassium) and most s-block and p-block elements don’t have partially filled d-orbitals. Without d-electrons that can jump between energy levels, d-d transitions can’t occur, leaving their solutions colourless.
Remember: No partially filled d-orbitals = No d-d transitions = No colour
This is why sodium chloride dissolves to give a completely clear solution, while copper sulfate gives that distinctive blue colour.
Exam Success Strategy: Master These Key Points ✅
For GCSE Students:
- Learn the colours of common transition metal ions
- Understand that d-electrons cause the colours
- Know that complex ions form when metals dissolve
- Practice identifying metals from their solution colours
For A Level Students:
- Understand crystal field splitting and d-d transitions
- Learn how ligands, oxidation states, and geometry affect colour
- Master the relationship between absorbed and observed colours
- Connect this to practical applications and industrial uses

If you’re feeling overwhelmed by the complexity of transition metal chemistry, you’re not alone! Many students find this topic challenging because it connects atomic structure, bonding theory, and practical applications all in one go.
The key to success is building your understanding step by step. Start with the basic concept that d-electrons can absorb light, then gradually add in the factors that affect which colours are absorbed and observed.
Making Connections: Your Next Steps
Understanding transition metal colours opens doors to so many other areas of chemistry! Once you’ve mastered this topic, you’ll find it easier to understand:
- Catalysis and how transition metals speed up reactions
- Redox chemistry and electron transfer processes
- Coordination compounds and their shapes
- Industrial processes that rely on coloured indicators
For students looking to excel in chemistry, whether you’re aiming for top GCSE grades or preparing for competitive university applications, mastering transition metals is absolutely essential. The visual nature of this topic makes it perfect for understanding deeper chemical principles while having something concrete and colourful to remember! 🌟
Remember: every time you see a blue copper solution or notice rust forming, you’re witnessing the same d-d electron transitions that you’re studying in class. Chemistry really is everywhere around us!

Ready to turn your understanding of transition metals into exam success? Practice drawing d-orbital splitting diagrams, create your own colour reference cards, and always ask yourself: “What’s causing this particular colour?” With consistent practice and the right approach, this topic can become one of your strongest chemistry skills! 💪
Oxford-Educated Chemistry Specialist
With over 20 years of teaching experience at some of the UK’s top independent schools, I help ambitious students bridge the gap between hard work and top-tier results. I specialise in GCSE, A Level, and IB Chemistry tuition for students targeting Grade 9s and A*s. Based in the UK but working globally, I provide 1-1 online support for families in South and West London, Dubai, and Hong Kong, ensuring students are perfectly prepared for competitive medical applications and Oxbridge entries.
I’ve helped students achieve top grades from schools such as Alleyn’s, Dulwich College, Tonbridge, Sevenoaks, Brighton College, Wycombe Abbey, Caterham, St Paul’s, Dubai College, Dubai British School and Harrow International School Hong Kong.
Contact me archardchloe@gmail.com to discuss how I can help your child excel in Chemistry.
